Abstract
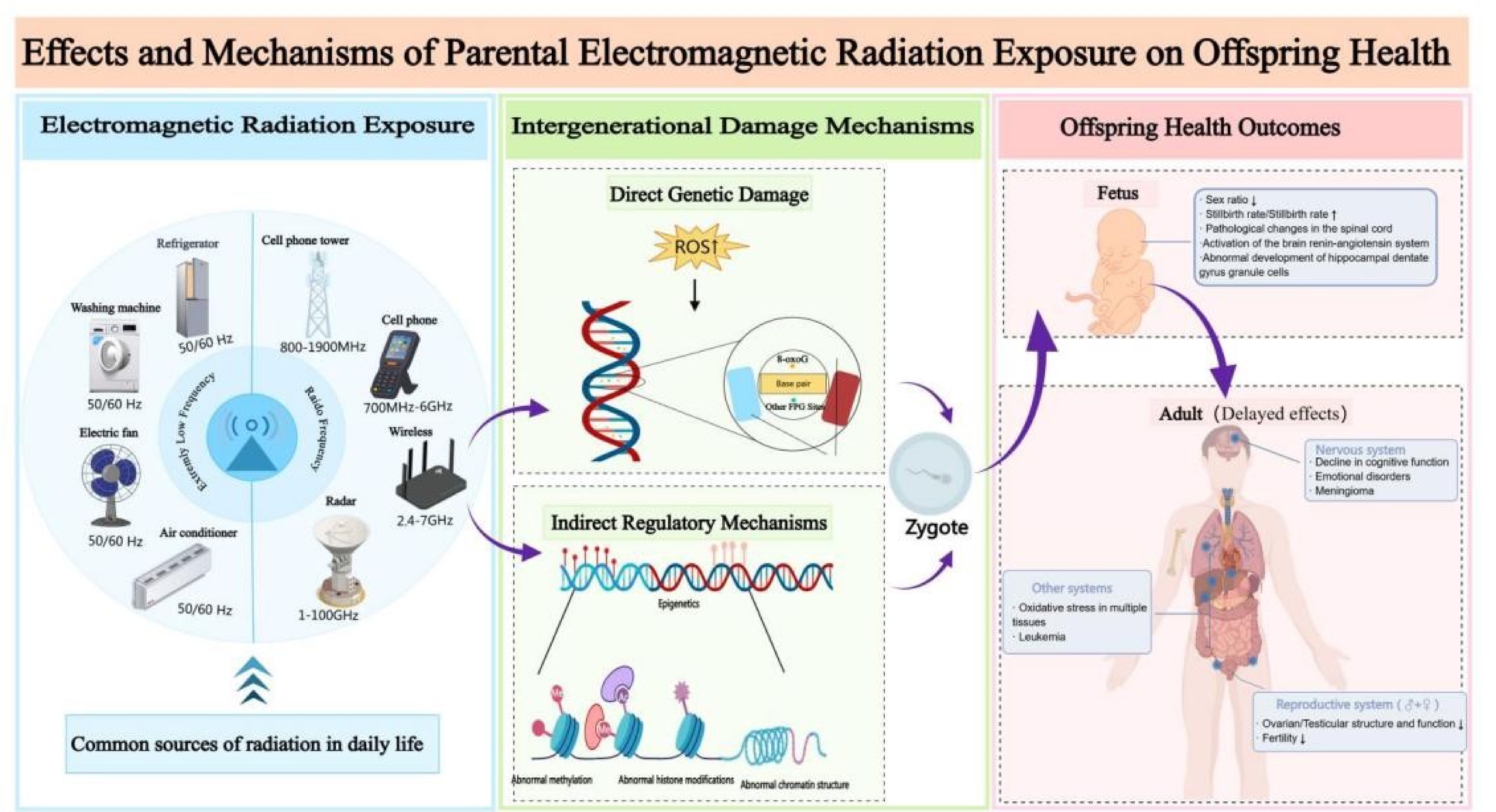
The advancement of technology has heightened concerns regarding the potential health effects of electromagnetic radiation (EMR) exposure on future generations. This review aims to synthesize current research on the health impacts of parental EMR exposure on offspring, focusing on underlying biological mechanisms and providing scientific evidence to support preventive health recommendations. Studies suggest parental EMR exposure may detrimentally impact offspring's neurological and reproductive systems. Numerous animal experiments and epidemiological studies indicate associations between EMR exposure and adverse outcomes, such as fetal dysplasia, cognitive impairment, and reproductive damage. This review further examines possible mechanisms, such as oxidative stress, apoptosis, and DNA damage. Investigating the health effects of EMR on offspring is crucial for developing preventive measures and intervention strategies to reduce congenital disabilities and enhance reproductive health. Given the methodological inconsistencies and variable results in existing studies, future research should focus on refining experimental designs to assess the long-term impacts of different EMR frequencies and intensities on developing individuals, thereby clarifying their public health implications. This review seeks to consolidate findings on the effects of parental EMR exposure on offspring health, elucidate underlying mechanisms, and provide evidence-based recommendations to support public health interventions for future generations.
Keywords:Electromagnetic radiation; Offspring; Nerve; Reproductive system;
Introduction
With technological advancements, the application of electromagnetic radiation in high-tech fields such as 5G networks and satellite communication is continuously expanding. This phenomenon has raised concerns about EMF (Electromagnetic Fields) exposure among the public.
Electromagnetic radiation is produced by the interaction of varying electric and magnetic fields, transmitting energy through space in waveform. Common examples include radio waves and microwaves, both of which are integral to daily life and technological applications. Electromagnetic radiation is broadly classified into radio frequency (RF) radiation (3 MHz to 300 GHz) and extremely low frequency (ELF) radiation (3 to 300 Hz), based on the frequency of the waves. The power density of electromagnetic radiation represents the amount of energy at a specific location, typically measured as power per unit area. This metric[1] , expressed in watts per square meter (W/m²), is used to quantify the strength or intensity of electromagnetic radiation. The unit of Specific Absorption Rate (SAR)[2] is watts per kilogram (W/kg), representing the electromagnetic radiation power absorbed by biological tissue per unit mass. SAR is commonly used to assess whether devices comply with international safety standards, with an average limit of 2.0 W/kg.
To assess the potential health impacts of long-term exposure to electromagnetic radiation in various populations, the World Health Organization (WHO) convened 300 experts[3] , who identified six priority areas of concern: cancer, heat-related effects, adverse reproductive outcomes, electromagnetic hypersensitivity, cognitive impairment, and oxidative stress. Adverse reproductive outcomes are considered one of the most critical factors influencing human development. During the embryonic development phase, embryos are highly susceptible to environmental influences. Electromagnetic waves can affect the developing embryo, potentially impacting fetal health. Studies have shown[4] that parental environmental exposure is one of the primary factors leading to congenital diseases in children. This review focuses on the health impacts of maternal and paternal electromagnetic radiation (EMR) exposure on offspring, with particular emphasis on underlying biological mechanisms and intergenerational effects. While maternal EMR exposure has been extensively investigated in fetal development research, paternal exposure remains significantly understudied. This asymmetry in scientific attention underscores a critical research gap: the lack of systematic comparisons between maternal and paternal EMR exposure in terms of biological mechanisms, critical sensitive windows, and long-term health outcomes. The present review aims to synthesize findings from epidemiological studies and animal experiments to compare the similarities and differences between maternal and paternal EMR exposure in mechanisms of action, critical exposure windows, and health consequences. Through systematic synthesis and comprehensive analysis, this study seeks to provide a scientific foundation for a holistic understanding of the potential risks posed by parental EMR exposure to offspring health, while offering insights to guide future research directions and inform public health intervention strategies.
Methods
Search strategy
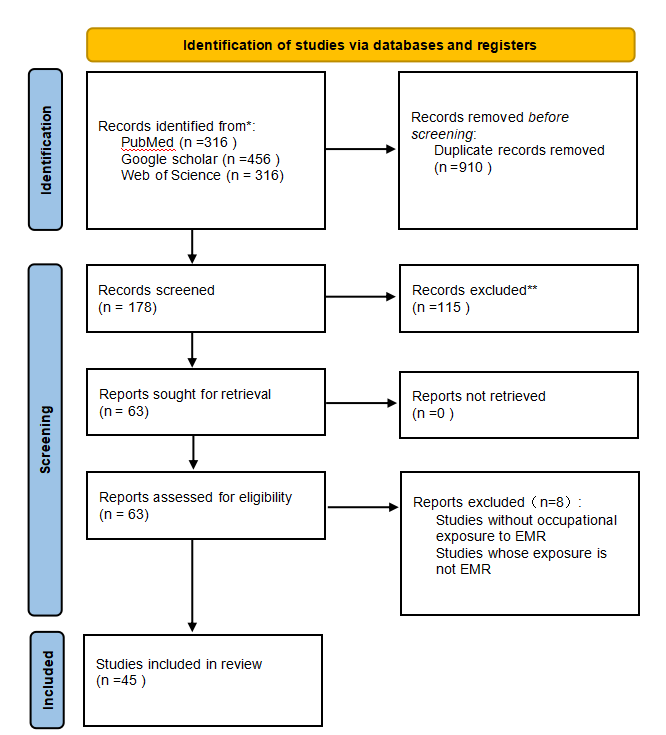
We developed this systematic review according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) recommendations. The search was performed using three different databases: PubMed, Google Scholar and Web of Science. To identify the publications eligible for inclusion we used the keywords “Electromagnetic radiation”; “Offspring”; “Nerve”; “Reproductive system” in the following search query: ((“electromagnetic radiation” AND “offspring” AND “parents”) AND “health”).Moreover, to limit the results, we applied the following research filters: articles published from 1 January 1990 to 1 August 2024, exclusion of animal studies and inclusion of publications in English only. As shown in Figure 1, an initial screening identified 70 candidate articles. After eliminating duplications, the initial screening of the studies was performed using information available in both the titles and the abstracts and 63 potentially relevant articles were retrieved in full text and assessed for eligibility according to the following inclusion criteria:Inclusion of both human and anima as study subject, with inclusion of offspring;Assessment of EMF exposure in parents;Inclusion of offspring health risks (For example, reproductive system damage, neurological damage, etc.)
The publications were included in the analysis only if they met all the eligibility criteria. Through the reading of the references of the included studies, after a full assessment of the potentially relevant studies, 45 articles met the eligibility criteria and were included in the systematic review.
Figure 1: PRISMA flow diagram.
Data extraction
During the analysis of the methodology and the results from the different studies, the authors noticed how these had been expressed either in a non-homogeneous or in a non-standardized manner. Therefore, the 45 articles in this systematic review were: 10 epidemiological studies, 35 animal experimentation. Relevant data were extracted, including
exposure time, source of exposure, frequency,average power density, SAR, intergenerationally inherited phenotype ,mechanism.
Methodological limitations
Given the substantial heterogeneity in animal study designs (e.g., species, exposure models, frequencies, and endpoints), we did not apply a single standardized risk of bias tool across all included studies. Instead, we evaluated study quality narratively, emphasizing reproducibility, exposure characterization, and biological plausibility of outcomes. Studies lacking basic methodological information (e.g., exposure parameters or offspring outcome data) were excluded to maintain baseline quality control.
Results and Discussion
Neurobehavioral development in offspring
Parental prenatal exposure to environmental EMF influences early embryonic neurological development, resulting in potential adverse effects on fetal growth and maturation. The brain, being one of the most sensitive organs to EMF exposure, may experience various types of neurological damage [5] , including neurological lesions, brain tumor formation, and cognitive and mood abnormalities. These adverse effects of electromagnetic radiation on the embryonic nervous system may potentially persist into postnatal life and adulthood. Some of these changes and the regulatory mechanisms involved are summarized in Table 1.
Abnormalities in nervous system development
Abnormal neurological development refers to deviations in the structural and functional formation of brain tissue during the embryonic stage. The development of the nervous system is a complex and highly coordinated process. Considering this complexity, numerous studies have focused on the potential effects of electromagnetic radiation exposure during critical developmental windows. Studies have investigated the effects of different durations of exposure to 900 MHz radiofrequency radiation during gestation, ranging from one hour to six hours per day. Ayşe İkinci Keleş et al. [6,7] found that exposure for one hour per day adversely impacted the morphological integrity of the spine and vertebrae in the offspring. Ersan Odaci et al.[8] found that the development of granule cells within the dentate gyrus of the hippocampus in rats was significantly affected by 900 MHz exposure. Aysu Kilic et al. [9] found that exposing both male and female rats to 900 MHz throughout the entire gestation period activated the brain renin-angiotensin system in the offspring, potentially leading to neuroinflammation and increased oxidative stress. Additionally, Haghani et al. [10] exposed pregnant rats to 900 MHz electromagnetic radiation for 6 hours per day, resulting in significant alterations in the electrophysiological properties of Purkinje neurons in the offspring. Several studies have explored the potential link between EMF exposure and neurological disorders. The mixed results from these studies highlight the complexity of the relationship between EMF exposure and nervous system development. While some studies indicate a potential risk, such as the association with meningiomas and central nervous system disorders, other studies fail to find significant evidence, suggesting that further research with more refined methodologies is needed to clarify these findings.
Neurological lesions
EMF can cause neurodegenerative syndromes and, in severe cases, may lead to diseases such as Parkinson's disease and Alzheimer's disease [11] . An epidemiological study conducted in Norway used data from 1967 to 1995. The study investigated the exposure of parents of 1.2 million newborns to electromagnetic fields. It found an association between potential central nervous system disorders and parental exposure to a 50 Hz magnetic field. [12] . A case-control study conducted in southwest France between May 1999 and April 2001 found that occupational or residential exposure to extremely low-frequency (ELF) EMFs may be associated with an increased risk of meningiomas[13] . However, a study by A.J. De Roos et al. [14] found no statistically significant association between the incidence of neuroblastoma in 538 children diagnosed in the USA or Canada between 1992 and 1994 and parental exposure to very low-frequency EMFs. A potential reason for these differing results could be the varying timing of exposure during critical periods of neurodevelopment. For example, prenatal exposure to electromagnetic fields may have a greater impact on fetal brain development, potentially leading to meningiomas or central nervous system disorders. In contrast, the relationship between electromagnetic fields and childhood neuroblastoma appears less clear, suggesting that certain types of EMF exposure may have a more limited effect on the development of specific childhood cancers.
Neurocognitive disorder
Cognitive impairment refers to a pathological condition characterized by abnormalities in the higher cognitive functions of the brain, such as learning, memory, thinking, and judgment, ultimately leading to cognitive decline. Indicators of cognitive impairment include reduced learning ability, diminished spatial memory, and inattention. For instance, a study by Moazamehosadat et al. [15] found that daily exposure of pregnant rats to 900 MHz radiofrequency radiation from mobile phones impaired cognitive performance in their female offspring. Yanchun Zhang et al. [16] exposed rats to high-frequency microwaves at 9.417 GHz, which are commonly used in certain industrial and communication devices, throughout gestation, resulting in gender-dependent learning and memory deficits, with males exhibiting more pronounced impairments compared to females.
An epidemiological study [17] utilizing data from 83,884 mother-child pairs across five countries provided a large-scale assessment of the potential effects of maternal mobile phone use during pregnancy, suggesting an association with an increased risk of behavioral problems in offspring, particularly hyperactivity/inattention problems. However, another experiment [18] exposed pregnant rats to 7.5 KHz electromagnetic radiation and did not observe significant impairments in cognitive function in the offspring. These discrepancies in findings may be attributed to differences in exposure parameters (e.g., frequency, power density), exposure duration, and species-specific responses, highlighting the complexity of assessing the effects of electromagnetic fields on neurodevelopment.
One possible explanation for the varying results could be differences in exposure parameters, such as frequency, power density, and duration, which may significantly influence the extent and nature of cognitive impairment. For example, high-frequency EMF exposure (e.g., 9.417 GHz) may have a more pronounced effect on brain development than low-frequency exposure (7.5 kHz), as higher frequencies penetrate tissues more effectively and may induce stronger biological effects. Furthermore, species-specific responses could contribute to some of the discrepancies observed, as different animal models may exhibit varying reactions to EMF exposure due to inherent differences in brain structure and metabolism. Gender differences in susceptibility might also be a factor; as demonstrated by Zhang et al. [16] , males exhibited more severe cognitive deficits than females, suggesting that hormonal or developmental differences could influence the response to EMFs.
Emotional disturbance
Regarding emotional disturbances, Burcu Acikgoz et al. [19] found that exposing pregnant rats to EMF was associated with increased anxiety-like behaviors in the offspring. Similarly, Yanchun Zhang et al. [16] demonstrated that exposure to 9.417 GHz during pregnancy was associated with heightened anxiety and reduced depressive-like behaviors in the offspring. Although these findings indicate possible links between EMF exposure and mood disturbances, relatively few studies have comprehensively explored mood disorders in offspring. Exposure to electromagnetic fields during pregnancy may interfere with maternal and fetal hormonal regulation, particularly hormones such as cortisol, oxytocin, and estrogen, which are involved in stress response and emotional processing. This disruption could contribute to emotional and behavioral problems in the offspring.
Table 1: The impact of parental electromagnetic radiation exposure on neurobehavioral development in offspring
|
Exposed Object |
Exposure Time/source of exposure |
Frequency |
Average Power Density |
SAR |
Intergenerationally Inherited Phenotype |
Main Mechanism |
Ref |
|
pregnant rat |
Exposure period: GD13.5-delivery Duration of exposure: 1h/day |
900MHz |
/ |
/ |
Pathological changes in the spinal cord |
epigenetic |
[6] |
|
pregnant rat |
Exposure period: GD23-PND 40 Duration of exposure: 1h/day |
900MHz |
/ |
/ |
activation of the brain renin-angiotensin system |
oxidative stress |
[9] |
|
pregnant rat |
Exposure period:13 to 21 days before delivery Duration of exposure: 1h/day |
900MHz |
/ |
/ |
Pathologic changes in the vertebrae |
/ |
[7] |
|
pregnant rat |
Exposure period: the entire pregnancy Duration of exposure: 1h/day |
900MHz |
1±0.4 W/cm2 |
2 W/kg |
abnormal development of hippocampal dentate gyrus granule cells |
Impaired cytokines |
[8] |
|
pregnant rat |
Exposure period: the entire pregnancy Duration of exposure: 6h/day |
900MHz |
/ |
0.5-0.9 W/kg |
the electrophysiological properties of Purkinje neurons are altered |
/ |
[10] |
|
pregnant rat |
Exposure period: the entire pregnancy |
900MHz |
/ |
0.3-0.9 W/kg |
decline in cognitive function |
/ |
[15] |
|
pregnant rat |
Exposure period: GD3.5-GD18 Duration of exposure:12h/day |
9.417GHz |
1.93 W/cm2 |
2 W/kg |
there are sex differences in emotional disorders and cognitive function |
/ |
[16] |
|
pregnant rat |
Exposure period: the entire pregnancy Duration of exposure: 24h/day |
7.5 kHz MF (12 or 120 μT) |
/ |
/ |
no effect |
/ |
[18] |
|
human parents |
Exposure period: occupational exposure |
50 Hz |
/ |
/ |
cause central nervous system diseases in offspring |
/ |
[12] |
|
human parents |
Exposure period: occupational exposure & residential exposure |
ELF |
/ |
/ |
cause meningioma in offspring |
/ |
[13] |
|
pregnant human mother |
Exposure period: the entire pregnancy |
radiofrequency radiation from mobile phones |
/ |
/ |
may cause behavioral disorders, particularly ADHD/inattention |
/ |
[17] |
|
human parents |
Exposure period: occupational exposure |
extremely low-frequency magnetic fields (>0.4 μT) |
/ |
/ |
There is no evidence that parental electromagnetic spectrum exposure |
/ |
[14] |
|
|
causes neuroblastoma in offspring |
Reproductive system damage in offspring.
Due to the unique structure and function of the reproductive system, it is particularly vulnerable to environmental factors, including EMF; therefore, parental exposure to EMF may negatively impact reproductive health and fertility by interfering with hormonal balance and gamete production[20] . Moreover, EMF exposure may affect not only the reproductive health of individuals but also the health of their offspring through genetic and epigenetic alterations. Table 2 summarizes some of these changes and the possible regulatory mechanisms involved.
Male reproductive system damage
Nevin Ersoy et al. [21] found that prenatal and postnatal exposure to 50-Hz, 3-mT EMF resulted in increased apoptosis and structural alterations in the testes of the offspring. Similarly, Alper Özorak et al. [22] exposed 32 pregnant mice to three frequencies—2.45 GHz, 900 MHz, and 1,800 MHz—and found that their offspring showed increased oxidative stress in testicular tissue and exhibited signs of precocious puberty, as indicated by accelerated sexual maturation. In contrast, Song Yan et al. [23] exposed male mice to 2.0 GHz RF EMR and did not observe any significant damage to the testicular structure or changes in sperm quality in the offspring. However, exposure of pregnant rats to 2.45 GHz RF EMR was reported [24] to have deleterious effects on postnatal testicular development, including impaired germ cell differentiation and disrupted spermatogenic process, which in turn impaired the spermatogenic cycle and male reproductive function. The studies reviewed demonstrate a range of effects of EMF exposure on male reproductive health, particularly with respect to testicular function and spermatogenesis in offspring. While some studies report significant damage, others find no observable harm, suggesting that the outcomes may be influenced by several factors, including variations in experimental design, species-specific responses, and differences in the timing and duration of exposure.
Female Reproductive System Injury
B. M. Ryan et al.[25] exposed F0 generation rats to EMF at 60 Hz for 17 weeks and found that parameters related to fertility, including litter size, gestational latency, and sex ratio, were not impaired in the F0, F1, and F2 generations. Acikgoz Burcu et al. [26] exposed rats to 50 Hz, 3 mT EMF both prenatally and postnatally, which was associated with impaired ovarian structure and function in female offspring, potentially through increased iNOS levels. According to report[27] , simultaneous whole-body exposure of pregnant rats to eight different communication signal EMFs, with frequencies ranging from 800 MHz to 5.2 GHz, did not result in any adverse effects on pregnancy or developmental outcomes in the F1 and F2 generations. Satoru Takahashi et al. [28] found that whole-body exposure to 2.14 GHz RF-EMR for 20 hours per day during gestation and lactation did not result in adverse effects on fertility, embryotoxicity, or teratogenicity in the F1 and F2 generations.A critical factor influencing the observed variability in outcomes is the timing of exposure. Studies that exposed rats to EMFs during specific developmental windows, such as gestation and early postnatal stages, may have led to more pronounced effects, particularly on the female reproductive system, as evidenced by Acikgoz Burcu et al. [26] . During these sensitive periods, hormonal and cellular processes essential for organ development, including those of the ovaries, may be more susceptible to disruption, which could explain the ovarian dysfunction observed in female offspring.
Damage to the human reproductive system
In recent years, environmental factors are increasingly recognized as having a significant detrimental impact on the reproductive health of human offspring. It has been reported [29] that occupational exposure to EMF from sources such as high-frequency antennas, communication equipment, substations, and radiolocation stations can alter the sex ratio of offspring, characterized by a decreased proportion of male infants and an increased proportion of female infants. N. Mageroy et al. [30] analyzed data from a cross-sectional study of 2,265 current employees of the Norwegian Navy. They found an increased risk of congenital anomalies and stillbirths among crew members serving on the KNM Kvikk following the installation of a 750 W high-frequency transmitter. Valborg Baste and colleagues conducted a cohort study [31] using data from the Norwegian Medical Birth Registry (singleton births, n = 37,920). The study found that acute paternal exposure to electromagnetic radiation was associated with an increased risk of perinatal death and pregnancies complicated by preeclampsia. Similarly, Valborg Baste [32] found that paternal exposure to mobile phone radiation during the six months prior to conception was associated with an increased risk of perinatal mortality, potentially due to alterations in sperm DNA integrity. A study[33] examined 53 workers who were occupationally exposed to electrical fields at a 400 kV substation for more than five years. The findings indicated that the workers had a relatively lower number of offspring compared to general population data, with a decreased proportion of male offspring. The evidence suggests that EMF exposure may have significant implications for reproductive health, particularly concerning male fertility and paternal effects on pregnancy outcomes. Notably, alterations in sex ratios and an increased incidence of perinatal complications associated with paternal EMF exposure raise concerns about the broader environmental impact on human reproduction. Given the growing prevalence of EMF exposure in contemporary society, it is imperative to investigate the underlying mechanisms that mediate these associations and to develop preventive strategies aimed at mitigating potential risks to future generations.
Table 2: The impact of parental electromagnetic radiation exposure on the reproductive system of offspring
|
Exposed Object |
Exposure Time/source of exposure |
Frequency |
Average Power Density |
SAR |
Intergenerationally Inherited Phenotype |
Mechanism |
Ref |
|
pregnant rat |
Exposure period:the entire pregnancy - PND 24/48 Duration of exposure: 4h/day 5day/week |
50Hz (3-mT) |
/ |
/ |
Testicular structural changes and germ cell apoptosis. |
epigenetic |
[21] |
|
pregnant rat &offspring |
Exposure period: GD7-postpartum Duration of exposure: 20h/day |
2.14GHz |
/ |
Female rat:0.066-0.093 W/kg offspring: 0.068-0.146 W/kg |
No effect |
/ |
[28] |
|
pregnant rat & offspring |
Exposure period: the entire pregnancy - PND 42 Duration of exposure: 1h/day 5day/week |
2.45 GHz、 900 MHz、 1800 MHz (single radiation exposure) |
0.12 W/m² |
0.18±0.07W/kg |
Precocious puberty and testicular oxidative damage |
/ |
[22] |
|
male mice |
Exposure period:14week Duration of exposure: 3h/day |
2.0 GHz |
2.5 W/m2 |
0.125-0.5 W/kg |
Body weight trajectory and glucose metabolism are altered in F1 male mice |
/ |
[23] |
|
pregnant rat & offspring |
Exposure period: the entire pregnancy - PND 63 Duration of exposure: 4h/day |
50 Hz (3 mT) |
/ |
/ |
Ovarian structure and function are impaired |
iNOS is activated |
[26] |
|
pregnant rat & offspring |
Exposure period: DG7 -PND 42 Duration of exposure: 20h/day |
800MHz、2GHz、2.4GHz、2.5GHz、5.2GHz (mixed radiation) |
/ |
0.08- 0.4W/kg |
Pregnancy and fertility are not impaired |
/ |
[27] |
|
pregnant rat |
Exposure period: the entire pregnancy Duration of exposure: 2h/day |
2.45GHz |
/ |
1.73 W/kg |
Male reproductive system is impaired |
/ |
[24] |
|
human parents |
Exposure period: occupational exposure |
high-frequency electromagnetic fields |
/ |
/ |
Congenital birth defects and stillbirth |
/ |
[30] |
|
human parents |
Exposure period: occupational exposure |
High-frequency antennas and communication devices, substations, physical therapists, radio navigation stations, etc. |
/ |
/ |
The proportion of male offspring is reduced |
/ |
[29] |
|
human father |
Exposure period: occupational exposure ①Acute exposure - within three months before pregnancy ②chronic exposure - more than three months before pregnancy |
2.1-4MHz、9.4GHz、9.1GHz |
/ |
/ |
Increased perinatal mortality rate |
/ |
[31] |
|
human father |
Exposure period: the six months before pregnancy |
radiofrequency radiation from mobile phones |
/ |
/ |
Increased perinatal mortality rate |
/ |
[32] |
Damage to other system functions in offspring
The effects of EMF on human health are not limited to the reproductive and nervous systems but may also extend to other vital organs, including the liver, heart, and kidneys (Table 3). For example, exposing pregnant rats to 900 MHz EMF for one hour per day resulted in oxidative stress and histopathological alterations in the liver of the offspring postnatally [34] . In addition to its effects on the liver, prolonged prenatal exposure of rats to high-frequency EMFs (900, 1800, and 2100 MHz) was associated with significant alterations in myocardial tissue structure and function [34,35] . A study [36] also investigated the effects of mobile phone (900-1800 MHz) induced electromagnetic radiation on the redox state of the heart, liver, kidney, cerebellum, and hippocampus in rats and their offspring, and they found that exposure to mobile phones during pregnancy induced oxidative stress in various tissues of the offspring, including the liver, kidneys, and heart.
Zozan Guleken et al.[37] investigated the long-term effects of extremely low-frequency magnetic fields (ELF-MF, 50 Hz), further highlighting the broad impacts of EMF exposure on biological systems. They showed that long-term EMF exposure is an environmental factor affecting serum parameters and may impair the oxidative-antioxidant balance of the organism. Short-term exposure to Wi-Fi-induced EMR before and after birth in rats may lead to an imbalance in tooth elemental composition, specifically affecting calcium and phosphorus levels, due to oxidative stress [38] . Alper Özorak et al. [39] exposed pregnant rats to three different frequencies (2.45 GHz, 900 MHz, and 1,800 MHz) and found that EMR led to nephrogenic oxidation in the growing offspring. Similarly, Joseph M. Lary et al. [40] reported that exposure of pregnant rats to a 27.12-MHz radiofrequency field caused fetal malformations, such as microphthalmia and brain enlargement, due to a radiation-induced increase in rectal temperature of 5°C, which may have contributed to these malformations.
Laboratories [41] have also used rabbits as research subjects, briefly exposing them to 1800 MHz radiation without finding significant pathological changes in the tissues and organs of the offspring. Furthermore, Mark S. Pearce et al. [42] analyzed data from the Northern Regional Registry of Malignant Diseases in the Young (NRYPMDR), comprising 4,723 cases, and found that occupational exposure to EMFs significantly increased the risk of developing leukemia, particularly acute lymphoblastic leukemia (ALL), in the male offspring. Similarly, Reid [43] found a moderate increase in the risk of acute lymphoblastic leukemia (ALL) in children whose mothers were occupationally exposed to extremely low-frequency (ELF) electromagnetic fields. These findings highlight the wide-ranging potential health risks associated with EMF exposure, including organ damage, developmental malformations, and an elevated risk of cancer. However, the variability in results across different studies necessitates further investigation to elucidate the underlying mechanisms, identify factors that influence susceptibility. A more comprehensive understanding of these issues is essential for accurately assessing the public health implications of EMFs in our increasingly interconnected and technology-driven society.
Table 3: The impact of parental electromagnetic radiation exposure on other tissues and organs in offspring
|
Exposed Object |
Exposure Time/source of exposure |
Frequency |
Average Power Density |
SAR |
Intergenerationally Inherited Phenotype |
Mechanism |
Ref |
|
Pregnant rat |
Exposure period: DG13-DG21 Duration of exposure: 1h/day |
900MHz |
0.54 W/m2 |
0.027 W/kg |
Oxidative stress and pathological changes occur in the liver |
/ |
[34] |
|
Pregnant rat |
Exposure period: 20day Duration of exposure:6h/day、12h/day、24h/day |
1800,2100,2450 MHz(single radiation exposure) |
/ |
0.087 W/kg,0.12 W/kg,0.17 W/kg |
Myocardial tissue is damaged |
/ |
[35] |
|
Pregnant mice |
Exposure period: 20day Duration of exposure:2h/day |
900-1800MHz |
/ |
/ |
Oxidative stress in various tissues and organs. |
/ |
[36] |
|
pregnant rat |
Exposure period: 42day [From pregnancy to lactation) Duration of exposure: 24h/day |
50 Hz (500μT) |
/ |
/ |
Affects serum parameters and increases lipid peroxidation and oxidative stress |
/ |
[37] |
|
pregnant rat |
Exposure period: 42day [From pregnancy to lactation) Duration of exposure: 2h/day |
2.45GHz |
/ |
0.009±0.002W/kg |
Imbalance in the oxidative stress status of the teeth |
/ |
[38] |
|
pregnant rat |
Exposure period: the entire pregnancy - PND 42 Duration of exposure: 1h/day 5day/week |
900 MHz,1800 MHz,2.45 GHz[single radiation exposure) |
0.12 W/m² |
0.18±0.07 W/kg |
Oxidative damage to the kidneys |
/ |
[22] |
|
pregnant rat |
Exposure period: DG9 Duration of exposure: 2h、15min |
27.12 MHz |
/ |
10.8 ± 0.3 W/kg |
Offspring malformations |
/ |
[40] |
|
pregnant rabbit |
Exposure period: 7day Duration of exposure: 15min/day |
1800MHz |
0.519 W/m2 |
/ |
No effect |
/ |
(46] |
|
human father |
Exposure period: occupational exposure |
power deviceshigh-frequency communication devicesradiological imaging equipment |
/ |
/ |
Increased risk of leukemia in male offspring |
/ |
[41] |
|
human mother |
Exposure period: occupational exposure |
ELE |
/ |
/ |
Increased risk of acute lymphoblastic leukemia in offspring |
/ |
(42] |
Conclusion
In summary, most recent animal studies and clinical evidence indicate that parental exposure to electromagnetic radiation, even at low doses, can adversely affect offspring health, impacting various organs and tissues, including the liver, brain, and reproductive system. Furthermore, long-term exposure to high-frequency radiation poses a greater hazard to offspring due to prenatal exposure compared to short-term exposure. However, a limited number of studies provide conflicting evidence, suggesting that under certain conditions, EMR exposure may not always lead to adverse health outcomes. These conflicting findings underscore the need for further well-controlled studies to clarify the conditions under which electromagnetic radiation might be harmful or benign to offspring health.
Existing studies on the health impacts of parental exposure to electromagnetic radiation (EMR) on offspring exhibit significant discrepancies, with contradictory findings attributable to the complex interplay of multidimensional factors. Firstly, inconsistencies in experimental design parameters represent a core cause. Variations in the selection of EMR frequency (e.g., 900 MHz vs. 7.5 kHz), intensity (differences in specific absorption rate (SAR) ranges), and exposure duration (1 to 24 hours/day) across studies could result in an undefined threshold for biological effects. High-frequency radiation, due to its stronger penetration ability, is more likely to induce oxidative stress or epigenetic alterations, whereas low-frequency or short-term exposure may obscure effects through biological adaptive mechanisms. Secondly, heterogeneity in study subjects and experimental models limits the comparability of outcomes. Physiological response differences between rodents and humans, strain-specific sensitivities in experimental animals (e.g., heightened hippocampal neuron vulnerability in rats compared to rabbits), and sex-specific responses (e.g., males being more prone to cognitive deficits) may all contribute to divergent conclusions. Thirdly, methodological limitations exacerbate uncertainty in results. Epidemiological studies relying on retrospective exposure assessments are prone to recall bias and susceptible to confounding factors (e.g., occupational chemical exposure), whereas animal experiments, despite controlled variables, fail to accurately replicate real-world multifrequency composite exposures and are limited by low statistical power in small-sample studies. Furthermore, diversity in biological endpoints and detection techniques (e.g., selection of oxidative stress markers, depth of epigenetic analyses) further complicates data interpretation.
The health effects of parental radiation exposure on offspring are complex, and only a small portion of the literature has explored the mechanisms of intergenerational inheritance, such as oxidative stress, iNOS activation, and epigenetic changes. However, these studies have primarily explored superficial mechanisms through animal experiments. Epigenetics, by contrast, provides a promising avenue for investigating deeper underlying mechanisms. Epigenetics is considered a form of non-genetic mechanism that regulates gene expression without altering the DNA sequence. A review [44] highlighted that environmental factors can induce behavioral changes in mammals, and these behavioral changes may be inherited by the next generation, thereby influencing the behavior of multiple generations. The external environment influences key carriers of epigenetic information, such as DNA methylation, histone modifications, and non-coding RNAs, which alters gene expression patterns and thus affects an individual's health [45] . These trait changes can not only persist throughout an individual's life but can sometimes be inherited across generations. With advancements in technology, the emergence of new techniques has provided powerful tools for studying the complex mechanisms of epigenetics, such as high-throughput sequencing technologies (e.g., Whole-Genome Bisulfite Sequencing and ChIP-seq), single-cell epigenetic technologies (e.g., scRNA-seq), and single-cell Hi-C technology [46] . These technologies help reveal how genes are regulated in different environments, developmental stages, and disease states
The current research literature on the effects of parental exposure to electromagnetic radiation (EMR) on offspring health is limited and faces several significant challenges. Firstly, the potential effects of EMR on offspring health may take years or even decades to manifest, making it difficult to conduct intergenerational genetic delayed-effect studies. Many contemporary studies do not include sufficiently long observation periods. Secondly, there is no consensus among researchers regarding standardized exposure conditions and outcome assessment methods, leading to inconsistencies in study conclusions. Additionally, more accurate simulations of exposure timing are needed in existing experiments. The health effects of EMR exposure on offspring may vary significantly depending on the timing, and real-life parental exposure often involves extended periods, including simultaneous postnatal exposure for both the offspring and the mother. This complexity makes accurate assessment and simulation of exposure conditions challenging. Moreover, species differences between humans and animal models, such as rats and rabbits, necessitate caution when extrapolating findings from animal experiments to clinical settings. Finally, mechanistic studies on intergenerational inheritance remain relatively unexplored, highlighting the need for further research using more sophisticated scientific methods to elucidate these mechanisms.
While existing research predominantly focuses on the direct impacts of maternal EMR exposure on embryonic development, paternal exposure has garnered increasing attention in recent years. This review reveals significant differences between maternal and paternal EMR exposure in mechanisms, sensitive windows, and health outcomes, as outlined below:
Divergent Mechanisms of Action
Maternal EMR exposure during pregnancy directly affects embryos through placental transmission, inducing oxidative stress, DNA damage, and epigenetic alterations [6,9] . For instance, gestational exposure of rats to 900 MHz EMR activates the renin-angiotensin system in offspring brains, leading to neuroinflammation [9] . In contrast, paternal EMR exposure primarily exerts its effects through compromised sperm DNA integrity or epigenetic modifications. Occupational paternal exposure to high-frequency EMR (e.g., 2.1–4 MHz) correlates with elevated perinatal mortality in offspring, likely mediated by sperm DNA fragmentation or mitochondrial dysfunction [31–32] .
Sensitivity of Exposure Windows
The embryonic period—particularly organogenesis—represents the critical window for maternal exposure. Studies indicate that maternal exposure to 9.417 GHz microwaves during early gestation (GD3.5–GD18) induces sex-dependent cognitive dysfunction in offspring [16] , whereas late-gestation exposure may provoke emotional disturbances [19] . Paternal sensitivity is concentrated during male gametogenesis (spermatogenesis). For example, frequent paternal mobile phone use within six months pre-conception is associated with increased perinatal mortality [32] , suggesting cumulative genetic damage during spermatogenesis.
Heterogeneity in Health Outcomes
Maternal EMR exposure is predominantly linked to offspring neurological abnormalities (e.g., cognitive deficits, anxiety-like behaviors) and structural malformations (e.g., spinal dysplasia) [6,16] . Epidemiological studies also suggest maternal mobile phone use during pregnancy may elevate offspring hyperactivity risk [17] . Paternal exposure, however, more profoundly impacts reproductive health and sex ratios. Occupational paternal exposure to high-frequency EMR (e.g., radar stations, communication devices) significantly reduces male offspring proportions [29] . Additionally, paternal exposure is associated with elevated leukemia risk in offspring [41–42] , potentially via germline mutations.
Systematic comparisons of paternal and maternal EMR exposure mechanisms and health effects are imperative to delineate pathway- and window-specific hazards, thereby informing targeted public health interventions.
Although existing evidence remains inconclusive, it suggests that adopting precautionary measures could mitigate potential risks. During critical windows of reproductive development (e.g., preconception, pregnancy, and early postnatal stages), exposure to high-frequency electromagnetic radiation (EMR) sources, such as 5G and mobile communication devices, should be minimized due to its established association with oxidative stress and epigenetic dysregulation in offspring. Occupational groups with high EMR exposure (e.g., telecommunications and healthcare workers) should prioritize protective strategies, including maintaining a safe distance from radiation sources and utilizing protective equipment. Regulatory frameworks should mandate standardized EMR dosimetry labeling on consumer electronic devices and adjust specific absorption rate (SAR) limits based on developmental stage sensitivity. Furthermore, public health communications should provide evidence-based guidance while avoiding the exaggeration of potential risks. However, these recommendations remain provisional—longitudinal studies investigating intergenerational epigenetic mechanisms are urgently required to address persistent uncertainties.
Overall, the evidence on the effects of EMR exposure on the health of offspring is still limited, and the underlying mechanisms remain poorly understood. As digital healthcare systems advance, it will become easier to conduct large-scale, longitudinal tracking of individual radiation exposure in the population, thereby enabling more precise investigations into the delayed effects of intergenerational inheritance. In addition, recent technological advancements enable the detection and quantification of DNA methylation, histone modifications, and RNA expression. These new approaches can also help uncover mechanisms of intergenerational inheritance from an epigenetic perspective or other relevant angles, and further identify reliable biomarkers of EMR exposure for future clinical diagnostics and treatments. This facilitates early intervention to prevent genetic diseases and birth defects, ultimately promoting healthy reproduction and childbearing.
Acknowledgements
Not applicable.
Funding information
This study is sponsored by Natural Science Foundation of Shanghai (24ZR1480000) and Shanghai Eastern Talent Plan (QNWS2024027).
Ethics approval and consent to participate
Ethical issues (Including plagiarism, informed consent, misconduct, data fabrication and/or falsification, double publication and/or submission, redundancy, etc.) have been completely observed by the authors.
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
References
[1] Muhibbullah M, Haleem AMA, Ikuma Y. (2017). Frequency dependent power and energy flux density equations of the electromagnetic wave. Results in Physics.7,435-9.https://doi.org/10.1016/j.rinp.2017.01.006
[2] Gautam R, Singh KV, Nirala J, Murmu NN, Meena R, Rajamani P. (2019). Oxidative stress-mediated alterations on sperm parameters in male Wistar rats exposed to 3G mobile phone radiation. Andrologia.51(3),e13201.https://doi.org/10.1111/and.13201
[3] Verbeek J, Oftedal G, Feychting M, van Rongen E, Rosaria Scarfì M, Mann S, et al. (2021). Prioritizing health outcomes when assessing the effects of exposure to radiofrequency electromagnetic fields: A survey among experts. Environment International.146,106300.https://doi.org/10.1016/j.envint.2020.106300
[4] Mahaldashtian M, Khalili MA, Anbari F, Seify M, Belli M. (2022). Challenges on the effect of cell phone radiation on mammalian embryos and fetuses: a review of the literature. Zygote.30(2),176-82.10.1017/S0967199421000691
[5] Lagorio S, Blettner M, Baaken D, Feychting M, Karipidis K, Loney T, et al. (2021). The effect of exposure to radiofrequency fields on cancer risk in the general and working population: A protocol for a systematic review of human observational studies. Environment International.157,106828.https://doi.org/10.1016/j.envint.2021.106828
[6] Keleş Aİ. (2020). Morphological changes in the vertebrae and central canal of rat pups born after exposure to the electromagnetic field of pregnant rats. Acta Histochemica.122(8),151652.https://doi.org/10.1016/j.acthis.2020.151652
[7] Keleş Aİ, Süt BB. (2021). Histopathological and epigenetic alterations in the spinal cord due to prenatal electromagnetic field exposure: An H3K27me3-related mechanism. Toxicology and Industrial Health.37(4),189-97.10.1177/0748233721996947
[8] Odaci E, Bas O, Kaplan S. (2008). Effects of prenatal exposure to a 900 MHz electromagnetic field on the dentate gyrus of rats: a stereological and histopathological study. Brain Research.1238,224-9.https://doi.org/10.1016/j.brainres.2008.08.013
[9] Kilic A, Ustunova S, Bulut H, Meral I. (2023). Pre and postnatal exposure to 900 MHz electromagnetic fields induce inflammation and oxidative stress, and alter renin-angiotensin system components differently in male and female offsprings. Life Sciences.321,121627.https://doi.org/10.1016/j.lfs.2023.121627
[10] Haghani M, Shabani M, Moazzami K. (2013). Maternal mobile phone exposure adversely affects the electrophysiological properties of Purkinje neurons in rat offspring. Neuroscience.250,588-98.https://doi.org/10.1016/j.neuroscience.2013.07.049
[11] Feychting M, Ahlbom A, Kheifets L. (2005). EMF and health. Annu Rev Public Health.26,165-89.10.1146/annurev.publhealth.26.021304.144445
[12] Blaasaas KG, Tynes T, Irgens A, Lie RT. (2002). Risk of birth defects by parental occupational exposure to 50 Hz electromagnetic fields: a population based study. Occup Environ Med.59(2),92-7.10.1136/oem.59.2.92
[13] Baldi I, Coureau G, Jaffré A, Gruber A, Ducamp S, Provost D, et al. (2011). Occupational and residential exposure to electromagnetic fields and risk of brain tumors in adults: a case-control study in Gironde, France. Int J Cancer.129(6),1477-84.10.1002/ijc.25765
[14] De Roos AJ, Teschke K, Savitz DA, Poole C, Grufferman S, Pollock BH, Olshan AF. (2001). Parental occupational exposures to electromagnetic fields and radiation and the incidence of neuroblastoma in offspring. Epidemiology.12(5),508-17.10.1097/00001648-200109000-00008
[15] Razavinasab M, Moazzami K, Shabani M. (2016). Maternal mobile phone exposure alters intrinsic electrophysiological properties of CA1 pyramidal neurons in rat offspring. Toxicol Ind Health.32(6),968-79.10.1177/0748233714525497
[16] Zhang Y, Li Z, Gao Y, Zhang C. (2015). Effects of fetal microwave radiation exposure on offspring behavior in mice. J Radiat Res.56(2),261-8.10.1093/jrr/rru097
[17] Birks L, Guxens M, Papadopoulou E, Alexander J, Ballester F, Estarlich M, et al. (2017). Maternal cell phone use during pregnancy and child behavioral problems in five birth cohorts. Environ Int.104,122-31.10.1016/j.envint.2017.03.024
[18] Kumari K, Koivisto H, Capstick M, Naarala J, Viluksela M, Tanila H, Juutilainen J. (2018). Behavioural phenotypes in mice after prenatal and early postnatal exposure to intermediate frequency magnetic fields. Environ Res.162,27-34.10.1016/j.envres.2017.12.013
[19] Acikgoz B, Ersoy N, Aksu I, Kiray A, Bagriyanik HA, Kiray M. (2022). Gender differences in effects of prenatal and postnatal exposure to electromagnetic field and prenatal zinc on behaviour and synaptic proteins in rats. J Chem Neuroanat.122,102092.10.1016/j.jchemneu.2022.102092
[20] Agarwal A, Singh A, Hamada A, Kesari K. (2011). Cell phones and male infertility: a review of recent innovations in technology and consequences. Int Braz J Urol.37(4),432-54.10.1590/s1677-55382011000400002
[21] Ersoy N, Acikgoz B, Aksu I, Kiray A, Bagriyanik HA, Kiray M. (2022). The Effects of Prenatal and Postnatal Exposure to 50-Hz and 3 mT Electromagnetic Field on Rat Testicular Development. Medicina (Kaunas).59(1).10.3390/medicina59010071
[22] Özorak A, Nazıroğlu M, Çelik Ö, Yüksel M, Özçelik D, Özkaya MO, et al. (2013). Wi-Fi (2.45 GHz)- and mobile phone (900 and 1800 MHz)-induced risks on oxidative stress and elements in kidney and testis of rats during pregnancy and the development of offspring. Biol Trace Elem Res.156(1-3),221-9.10.1007/s12011-013-9836-z
[23] Yan S, Ju Y, Dong J, Lei H, Wang J, Xu Q, et al. (2022). Paternal Radiofrequency Electromagnetic Radiation Exposure Causes Sex-Specific Differences in Body Weight Trajectory and Glucose Metabolism in Offspring Mice. Front Public Health.10,872198.10.3389/fpubh.2022.872198
[24] Andrašková S, Holovská K, Ševčíková Z, Andrejčáková Z, Tóth Š, Martončíková M, et al. (2022). The potential adverse effect of 2.45 GHz microwave radiation on the testes of prenatally exposed peripubertal male rats. Histol Histopathol.37(3),287-99.10.14670/hh-18-402
[25] Ryan BM, Symanski RR, Pomeranz LE, Johnson TR, Gauger JR, McCormick DL. (1999). Multigeneration reproductive toxicity assessment of 60-Hz magnetic fields using a continuous breeding protocol in rats. Teratology.59(3),156-62.10.1002/(sici)1096-9926(199903)59:3<156::Aid-tera7>3.0.Co;2-b
[26] Burcu A, Nevin E, Ilkay A, Amac K, Alper BH, Muge K. (2020). The effects of prenatal and postnatal exposure to electromagnetic field on rat ovarian tissue. Toxicology and Industrial Health.36(12),1010-8.10.1177/0748233720973136
[27] Shirai T, Wang J, Kawabe M, Wake K, Watanabe S-i, Takahashi S, Fujiwara O. (2017). No adverse effects detected for simultaneous whole-body exposure to multiple-frequency radiofrequency electromagnetic fields for rats in the intrauterine and pre- and post-weaning periods. Journal of Radiation Research.58(1),48-58.10.1093/jrr/rrw085
[28] Takahashi S, Imai N, Nabae K, Wake K, Kawai H, Wang J, et al. (2009). Lack of Adverse Effects of Whole-Body Exposure to a Mobile Telecommunication Electromagnetic Field on the Rat Fetus. Radiation Research.173(3),362-72.10.1667/rr1615.1
[29] Baste V, Riise T, Moen BE. (2008). Radiofrequency electromagnetic fields; male infertility and sex ratio of offspring. European Journal of Epidemiology.23(5),369-77.10.1007/s10654-008-9236-4
[30] Mageroy N, Mollerlokken OJ, Riise T, Koefoed V, Moen BE. (2006). A higher risk of congenital anomalies in the offspring of personnel who served aboard a Norwegian missile torpedo boat. Occupational and Environmental Medicine.63(2),92.10.1136/oem.2005.021113
[31] Baste V, Moen BE, Oftedal G, Strand LÅ, Bjørge L, Mild KH. (2012). Pregnancy Outcomes After Paternal Radiofrequency Field Exposure Aboard Fast Patrol Boats. Journal of Occupational and Environmental Medicine.54(4),431-8.10.1097/JOM.0b013e3182445003
[32] Baste V, Oftedal G, Møllerløkken OJ, Hansson Mild K, Moen BE. (2015). Prospective Study of Pregnancy Outcomes After Parental Cell Phone Exposure: The Norwegian Mother and Child Cohort Study. Epidemiology.26(4),613-21.10.1097/ede.0000000000000293
[33] Knave B, Gamberale F, BergstrÖM S, Birke E, Iregren A, Kolmodin-Hedman B, Wennberg A. (1979). Long-term exposure to electric fields: A cross-sectional epidemiologic investigation of occupationally exposed workers in high-voltage substations. Scandinavian Journal of Work, Environment & Health.5(2),115-25
[34] Topal Z, Hanci H, Mercantepe T, Erol HS, Keleş ON, Kaya H, et al. (2015). The effects of prenatal long-duration exposure to 900-MHz electromagnetic field on the 21-day-old newborn male rat liver. Turk J Med Sci.45(2),291-7.10.3906/sag-1404-168
[35] Bozok S, Karaagac E, Sener D, Akakin D, Tumkaya L. (2022). The effects of long-term prenatal exposure to 900, 1800, and 2100 MHz electromagnetic field radiation on myocardial tissue of rats. Toxicology and Industrial Health.39(1),1-9.10.1177/07482337221139586
[36] Bahreyni Toossi MH, Reza SH, Maryam MMF, Mahmoud H, Mahdiyeh H, Razieh M, et al. (2018). Exposure to mobile phone (900–1800 MHz) during pregnancy: tissue oxidative stress after childbirth. The Journal of Maternal-Fetal & Neonatal Medicine.31(10),1298-303.10.1080/14767058.2017.1315657
[37] Guleken Z, Sarıbal D, Uyulan C, Keles A, Depciuch J. (2021). Investigating Bio-interface Effects of Chronic ELF-MF Exposure before and after Neonatal Life on Rat Offspring Using Spectroscopy and Biochemical Assays. Biointerface Research in Applied Chemistry.12(1),795 - 808.https://doi.org/10.33263/BRIAC121.795808
[38] Çiftçi ZZ, Kırzıoğlu Z, Nazıroğlu M, Özmen Ö. (2015). Effects of Prenatal and Postnatal Exposure of Wi-Fi on Development of Teeth and Changes in Teeth Element Concentration in Rats. Biological Trace Element Research.163(1),193-201.10.1007/s12011-014-0175-5
[39] Özorak A, Nazıroğlu M, Çelik Ö, Yüksel M, Özçelik D, Özkaya MO, et al. (2013). Wi-Fi (2.45 GHz)- and Mobile Phone (900 and 1800 MHz)-Induced Risks on Oxidative Stress and Elements in Kidney and Testis of Rats During Pregnancy and the Development of Offspring. Biological Trace Element Research.156(1),221-9.10.1007/s12011-013-9836-z
[40] Lary JM, Conover DL, Johnson PH, Burg JR. (1983). Teratogenicity of 27.12-MHz radiation in rats is related to duration of hyperthermic exposure. Bioelectromagnetics.4(3),249-55.https://doi.org/10.1002/bem.2250040305
[41] GÜLER ÖZTÜRK G, Tomruk A, Ozgur E, Seyhan N. (2010). The effect of radiofrequency radiation on DNA and lipid damage in non-pregnant and pregnant rabbits and their newborns. General physiology and biophysics.29(1).https://10.4149/gpb_2010_01_59
[42] Pearce MS, Hammal DM, Dorak MT, McNally RJQ, Parker L. (2007). Paternal occupational exposure to electro-magnetic fields as a risk factor for cancer in children and young adults: A case-control study from the North of England. Pediatric Blood & Cancer.49(3),280-6.https://doi.org/10.1002/pbc.21021
[43] Reid A, Glass DC, Bailey HD, Milne E, de Klerk NH, Downie P, et al. (2011). Risk of childhood acute lymphoblastic leukaemia following parental occupational exposure to extremely low frequency electromagnetic fields. British Journal of Cancer.105(9),1409-13.10.1038/bjc.2011.365
[44] Bohacek J, Mansuy IM. (2015). Molecular insights into transgenerational non-genetic inheritance of acquired behaviours. Nature Reviews Genetics.16(11),641-52.10.1038/nrg3964
[45] Cavalli G, Heard E. (2019). Advances in epigenetics link genetics to the environment and disease. Nature.571(7766),489-99.10.1038/s41586-019-1411-0
[46] Nagano T, Lubling Y, Stevens TJ, Schoenfelder S, Yaffe E, Dean W, et al. (2013). Single-cell Hi-C reveals cell-to-cell variability in chromosome structure. Nature.502(7469),59-64.10.1038/nature12593
 Figures
Figures References
References Peer
Peer Information
InformationFigure 1: PRISMA flow diagram.

[1] Muhibbullah M, Haleem AMA, Ikuma Y. (2017). Frequency dependent power and energy flux density equations of the electromagnetic wave. Results in Physics.7,435-9.https://doi.org/10.1016/j.rinp.2017.01.006
[2] Gautam R, Singh KV, Nirala J, Murmu NN, Meena R, Rajamani P. (2019). Oxidative stress-mediated alterations on sperm parameters in male Wistar rats exposed to 3G mobile phone radiation. Andrologia.51(3),e13201.https://doi.org/10.1111/and.13201
[3] Verbeek J, Oftedal G, Feychting M, van Rongen E, Rosaria Scarfì M, Mann S, et al. (2021). Prioritizing health outcomes when assessing the effects of exposure to radiofrequency electromagnetic fields: A survey among experts. Environment International.146,106300.https://doi.org/10.1016/j.envint.2020.106300
[4] Mahaldashtian M, Khalili MA, Anbari F, Seify M, Belli M. (2022). Challenges on the effect of cell phone radiation on mammalian embryos and fetuses: a review of the literature. Zygote.30(2),176-82.10.1017/S0967199421000691
[5] Lagorio S, Blettner M, Baaken D, Feychting M, Karipidis K, Loney T, et al. (2021). The effect of exposure to radiofrequency fields on cancer risk in the general and working population: A protocol for a systematic review of human observational studies. Environment International.157,106828.https://doi.org/10.1016/j.envint.2021.106828
[6] Keleş Aİ. (2020). Morphological changes in the vertebrae and central canal of rat pups born after exposure to the electromagnetic field of pregnant rats. Acta Histochemica.122(8),151652.https://doi.org/10.1016/j.acthis.2020.151652
[7] Keleş Aİ, Süt BB. (2021). Histopathological and epigenetic alterations in the spinal cord due to prenatal electromagnetic field exposure: An H3K27me3-related mechanism. Toxicology and Industrial Health.37(4),189-97.10.1177/0748233721996947
[8] Odaci E, Bas O, Kaplan S. (2008). Effects of prenatal exposure to a 900 MHz electromagnetic field on the dentate gyrus of rats: a stereological and histopathological study. Brain Research.1238,224-9.https://doi.org/10.1016/j.brainres.2008.08.013
[9] Kilic A, Ustunova S, Bulut H, Meral I. (2023). Pre and postnatal exposure to 900 MHz electromagnetic fields induce inflammation and oxidative stress, and alter renin-angiotensin system components differently in male and female offsprings. Life Sciences.321,121627.https://doi.org/10.1016/j.lfs.2023.121627
[10] Haghani M, Shabani M, Moazzami K. (2013). Maternal mobile phone exposure adversely affects the electrophysiological properties of Purkinje neurons in rat offspring. Neuroscience.250,588-98.https://doi.org/10.1016/j.neuroscience.2013.07.049
[11] Feychting M, Ahlbom A, Kheifets L. (2005). EMF and health. Annu Rev Public Health.26,165-89.10.1146/annurev.publhealth.26.021304.144445
[12] Blaasaas KG, Tynes T, Irgens A, Lie RT. (2002). Risk of birth defects by parental occupational exposure to 50 Hz electromagnetic fields: a population based study. Occup Environ Med.59(2),92-7.10.1136/oem.59.2.92
[13] Baldi I, Coureau G, Jaffré A, Gruber A, Ducamp S, Provost D, et al. (2011). Occupational and residential exposure to electromagnetic fields and risk of brain tumors in adults: a case-control study in Gironde, France. Int J Cancer.129(6),1477-84.10.1002/ijc.25765
[14] De Roos AJ, Teschke K, Savitz DA, Poole C, Grufferman S, Pollock BH, Olshan AF. (2001). Parental occupational exposures to electromagnetic fields and radiation and the incidence of neuroblastoma in offspring. Epidemiology.12(5),508-17.10.1097/00001648-200109000-00008
[15] Razavinasab M, Moazzami K, Shabani M. (2016). Maternal mobile phone exposure alters intrinsic electrophysiological properties of CA1 pyramidal neurons in rat offspring. Toxicol Ind Health.32(6),968-79.10.1177/0748233714525497
[16] Zhang Y, Li Z, Gao Y, Zhang C. (2015). Effects of fetal microwave radiation exposure on offspring behavior in mice. J Radiat Res.56(2),261-8.10.1093/jrr/rru097
[17] Birks L, Guxens M, Papadopoulou E, Alexander J, Ballester F, Estarlich M, et al. (2017). Maternal cell phone use during pregnancy and child behavioral problems in five birth cohorts. Environ Int.104,122-31.10.1016/j.envint.2017.03.024
[18] Kumari K, Koivisto H, Capstick M, Naarala J, Viluksela M, Tanila H, Juutilainen J. (2018). Behavioural phenotypes in mice after prenatal and early postnatal exposure to intermediate frequency magnetic fields. Environ Res.162,27-34.10.1016/j.envres.2017.12.013
[19] Acikgoz B, Ersoy N, Aksu I, Kiray A, Bagriyanik HA, Kiray M. (2022). Gender differences in effects of prenatal and postnatal exposure to electromagnetic field and prenatal zinc on behaviour and synaptic proteins in rats. J Chem Neuroanat.122,102092.10.1016/j.jchemneu.2022.102092
[20] Agarwal A, Singh A, Hamada A, Kesari K. (2011). Cell phones and male infertility: a review of recent innovations in technology and consequences. Int Braz J Urol.37(4),432-54.10.1590/s1677-55382011000400002
[21] Ersoy N, Acikgoz B, Aksu I, Kiray A, Bagriyanik HA, Kiray M. (2022). The Effects of Prenatal and Postnatal Exposure to 50-Hz and 3 mT Electromagnetic Field on Rat Testicular Development. Medicina (Kaunas).59(1).10.3390/medicina59010071
[22] Özorak A, Nazıroğlu M, Çelik Ö, Yüksel M, Özçelik D, Özkaya MO, et al. (2013). Wi-Fi (2.45 GHz)- and mobile phone (900 and 1800 MHz)-induced risks on oxidative stress and elements in kidney and testis of rats during pregnancy and the development of offspring. Biol Trace Elem Res.156(1-3),221-9.10.1007/s12011-013-9836-z
[23] Yan S, Ju Y, Dong J, Lei H, Wang J, Xu Q, et al. (2022). Paternal Radiofrequency Electromagnetic Radiation Exposure Causes Sex-Specific Differences in Body Weight Trajectory and Glucose Metabolism in Offspring Mice. Front Public Health.10,872198.10.3389/fpubh.2022.872198
[24] Andrašková S, Holovská K, Ševčíková Z, Andrejčáková Z, Tóth Š, Martončíková M, et al. (2022). The potential adverse effect of 2.45 GHz microwave radiation on the testes of prenatally exposed peripubertal male rats. Histol Histopathol.37(3),287-99.10.14670/hh-18-402
[25] Ryan BM, Symanski RR, Pomeranz LE, Johnson TR, Gauger JR, McCormick DL. (1999). Multigeneration reproductive toxicity assessment of 60-Hz magnetic fields using a continuous breeding protocol in rats. Teratology.59(3),156-62.10.1002/(sici)1096-9926(199903)59:3<156::Aid-tera7>3.0.Co;2-b
[26] Burcu A, Nevin E, Ilkay A, Amac K, Alper BH, Muge K. (2020). The effects of prenatal and postnatal exposure to electromagnetic field on rat ovarian tissue. Toxicology and Industrial Health.36(12),1010-8.10.1177/0748233720973136
[27] Shirai T, Wang J, Kawabe M, Wake K, Watanabe S-i, Takahashi S, Fujiwara O. (2017). No adverse effects detected for simultaneous whole-body exposure to multiple-frequency radiofrequency electromagnetic fields for rats in the intrauterine and pre- and post-weaning periods. Journal of Radiation Research.58(1),48-58.10.1093/jrr/rrw085
[28] Takahashi S, Imai N, Nabae K, Wake K, Kawai H, Wang J, et al. (2009). Lack of Adverse Effects of Whole-Body Exposure to a Mobile Telecommunication Electromagnetic Field on the Rat Fetus. Radiation Research.173(3),362-72.10.1667/rr1615.1
[29] Baste V, Riise T, Moen BE. (2008). Radiofrequency electromagnetic fields; male infertility and sex ratio of offspring. European Journal of Epidemiology.23(5),369-77.10.1007/s10654-008-9236-4
[30] Mageroy N, Mollerlokken OJ, Riise T, Koefoed V, Moen BE. (2006). A higher risk of congenital anomalies in the offspring of personnel who served aboard a Norwegian missile torpedo boat. Occupational and Environmental Medicine.63(2),92.10.1136/oem.2005.021113
[31] Baste V, Moen BE, Oftedal G, Strand LÅ, Bjørge L, Mild KH. (2012). Pregnancy Outcomes After Paternal Radiofrequency Field Exposure Aboard Fast Patrol Boats. Journal of Occupational and Environmental Medicine.54(4),431-8.10.1097/JOM.0b013e3182445003
[32] Baste V, Oftedal G, Møllerløkken OJ, Hansson Mild K, Moen BE. (2015). Prospective Study of Pregnancy Outcomes After Parental Cell Phone Exposure: The Norwegian Mother and Child Cohort Study. Epidemiology.26(4),613-21.10.1097/ede.0000000000000293
[33] Knave B, Gamberale F, BergstrÖM S, Birke E, Iregren A, Kolmodin-Hedman B, Wennberg A. (1979). Long-term exposure to electric fields: A cross-sectional epidemiologic investigation of occupationally exposed workers in high-voltage substations. Scandinavian Journal of Work, Environment & Health.5(2),115-25
[34] Topal Z, Hanci H, Mercantepe T, Erol HS, Keleş ON, Kaya H, et al. (2015). The effects of prenatal long-duration exposure to 900-MHz electromagnetic field on the 21-day-old newborn male rat liver. Turk J Med Sci.45(2),291-7.10.3906/sag-1404-168
[35] Bozok S, Karaagac E, Sener D, Akakin D, Tumkaya L. (2022). The effects of long-term prenatal exposure to 900, 1800, and 2100 MHz electromagnetic field radiation on myocardial tissue of rats. Toxicology and Industrial Health.39(1),1-9.10.1177/07482337221139586
[36] Bahreyni Toossi MH, Reza SH, Maryam MMF, Mahmoud H, Mahdiyeh H, Razieh M, et al. (2018). Exposure to mobile phone (900–1800 MHz) during pregnancy: tissue oxidative stress after childbirth. The Journal of Maternal-Fetal & Neonatal Medicine.31(10),1298-303.10.1080/14767058.2017.1315657
[37] Guleken Z, Sarıbal D, Uyulan C, Keles A, Depciuch J. (2021). Investigating Bio-interface Effects of Chronic ELF-MF Exposure before and after Neonatal Life on Rat Offspring Using Spectroscopy and Biochemical Assays. Biointerface Research in Applied Chemistry.12(1),795 - 808.https://doi.org/10.33263/BRIAC121.795808
[38] Çiftçi ZZ, Kırzıoğlu Z, Nazıroğlu M, Özmen Ö. (2015). Effects of Prenatal and Postnatal Exposure of Wi-Fi on Development of Teeth and Changes in Teeth Element Concentration in Rats. Biological Trace Element Research.163(1),193-201.10.1007/s12011-014-0175-5
[39] Özorak A, Nazıroğlu M, Çelik Ö, Yüksel M, Özçelik D, Özkaya MO, et al. (2013). Wi-Fi (2.45 GHz)- and Mobile Phone (900 and 1800 MHz)-Induced Risks on Oxidative Stress and Elements in Kidney and Testis of Rats During Pregnancy and the Development of Offspring. Biological Trace Element Research.156(1),221-9.10.1007/s12011-013-9836-z
[40] Lary JM, Conover DL, Johnson PH, Burg JR. (1983). Teratogenicity of 27.12-MHz radiation in rats is related to duration of hyperthermic exposure. Bioelectromagnetics.4(3),249-55.https://doi.org/10.1002/bem.2250040305
[41] GÜLER ÖZTÜRK G, Tomruk A, Ozgur E, Seyhan N. (2010). The effect of radiofrequency radiation on DNA and lipid damage in non-pregnant and pregnant rabbits and their newborns. General physiology and biophysics.29(1).https://10.4149/gpb_2010_01_59
[42] Pearce MS, Hammal DM, Dorak MT, McNally RJQ, Parker L. (2007). Paternal occupational exposure to electro-magnetic fields as a risk factor for cancer in children and young adults: A case-control study from the North of England. Pediatric Blood & Cancer.49(3),280-6.https://doi.org/10.1002/pbc.21021
[43] Reid A, Glass DC, Bailey HD, Milne E, de Klerk NH, Downie P, et al. (2011). Risk of childhood acute lymphoblastic leukaemia following parental occupational exposure to extremely low frequency electromagnetic fields. British Journal of Cancer.105(9),1409-13.10.1038/bjc.2011.365
[44] Bohacek J, Mansuy IM. (2015). Molecular insights into transgenerational non-genetic inheritance of acquired behaviours. Nature Reviews Genetics.16(11),641-52.10.1038/nrg3964
[45] Cavalli G, Heard E. (2019). Advances in epigenetics link genetics to the environment and disease. Nature.571(7766),489-99.10.1038/s41586-019-1411-0
[46] Nagano T, Lubling Y, Stevens TJ, Schoenfelder S, Yaffe E, Dean W, et al. (2013). Single-cell Hi-C reveals cell-to-cell variability in chromosome structure. Nature.502(7469),59-64.10.1038/nature12593
Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-01-22
Accepted 2025-02-17
Published 2025-03-30


