Abstract

Pulmonary arterial hypertension (PAH) is a life-threatening progressive disorder caused by increased resistance in the pulmonary vasculature. The condition may lead to right-sided heart failure and death. A 56-year-old woman was referred for pulmonary arterial hypertension (PAH) with atrial septal defect, mitral regurgitation, and tricuspid regurgitation. Hemodynamic parameters were obtained by right heart catheterization. She underwent transthoracic pulmonary artery radiofrequency denervation (TPARFD), to relieve the pulmonary artery pressure, while concomitantly undergoing mitral valvuloplasty with 28 # Sorin ring, patch closure of atrial septal defect, and tricuspid annuloplasty with 28 # Edwards MC3 ring. Her hemodynamic parameters and functional capacity showed marked improvement after TPARFD.
Keywords:Pulmonary artery radiofrequency denervation; Pulmonary arterial hypertension; Congenital heart disease.
Introduction
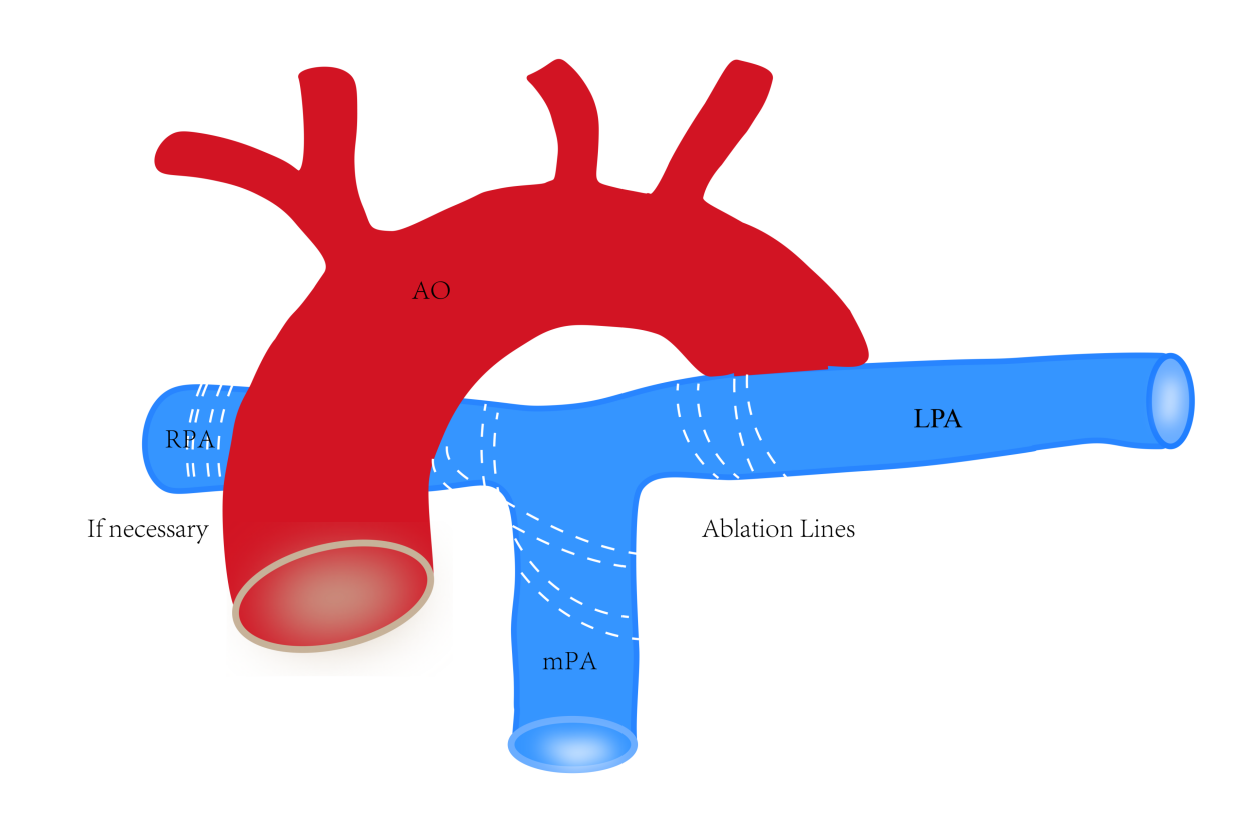
Pulmonary arterial hypertension (PAH) is a life-threatening progressive disorder. The high prevalence of pulmonary arterial hypertension in adults is commonly associated with congenital heart disease [1] . A majority of the cardiac defects are successfully corrected by surgery; however, a considerable proportion of patients experience aggravation of PAH and remain at a high risk [2] . Several clinical studies have demonstrated the safety and effectiveness of percutaneous pulmonary artery denervation (PADN). However, pre-operative patients with congenital heart disease were not included in the clinical studies [3-5] . In addition, PAND alone, without repairing the simple pre- or post-tricuspid shunts, might insult the pulmonary pulmonary circulation in the PAH-CHD patients. Herein, we designed a new surgical operation method, transthoracic pulmonary artery radiofrequency denervation (TPARFD), to concomitantly relieve the pulmonary artery pressure during repair of the pre- or post-tricuspid shunts in PAH-ACHD (Fig. 1).
Case Description
A 56-year-old woman was diagnosed with atrial septal defect seven months ago. The patient was asymptomatic and did not receive any treatment. On February 22, 2021, the patient came to our hospital for further treatment. At admission, her blood pressure was 125/68 mmHg and her pulse rate was 70 bpm. The New York Heart Association functional classification was class III. Six-minute walk distance was 300 m. The B-type natriuretic peptide (BNP) level was 19.8 pg/mL. Transthoracic echocardiography revealed an atrial septal defect (size: 35.9×35.6 mm) with left to right shunt, marked enlargement of the right atrium (size: 53×65.5 mm), left ventricle end-diastolic dimension of 40 mm, ejection fraction of 66%, tricuspid regurgitation, severe PH with a tricuspid systolic pressure gradient of 82 mmHg. Tricuspid Annular Plane Systolic Excursion (TAPSE) was 23.5 mm. Right ventricular fractional area of change (RV-FAC) was 47%. The diameter of the central pulmonary artery was 43.5 mm. Twelve-lead electrocardiogram showed sinus rhythm, complete right bundle branch block, and QRS wave width of 114 ms. Right heart catheterization (RHC) showed elevated mean pulmonary arterial pressure (mPAP) and pulmonary vascular resistance (PVR) of 40 mmHg and 4.45 Wood, respectively. Cardiac output was 8.31 L/min estimated by the Fick method. The Qp/Qs ratio was 1.85. We started supplemental oxygen therapy and medical therapy with ambrisentan (10mg/day) and sildenafil (75mg/day).
She underwent transthoracic pulmonary artery radiofrequency denervation (TPARFD) to relieve the pulmonary artery pressure, while concomitantly undergoing mitral valvuloplasty with 28 # sorin ring, patch closure of atrial septal defect, and tricuspid annuloplasty with 28 # Edwards MC3 ring.
The combination medical therapy was continued after procedure. Six-minute walk distance was increased to 350 m at 1 month and 420 m at 3 months. The New York Heart Association functional classification was class II without oxygen administration, and the chest X-ray and echocardiography demonstrated the improvement of right ventricular overload. Her BNP level was 98.8 pg/mL. RHC performed 3 months after operation showed marked improvement in cardiac hemodynamics 3 days after stopping taking medications, mPAP was 19 mmHg and PVR was 2.55 Wood units. There was no airway injury, hemoptysis, PA dissection, vessel defect, pulmonary embolism, or hemorrhagic pleural effusion.
Transthoracic Pulmonary Artery Radiofreqency Denervation
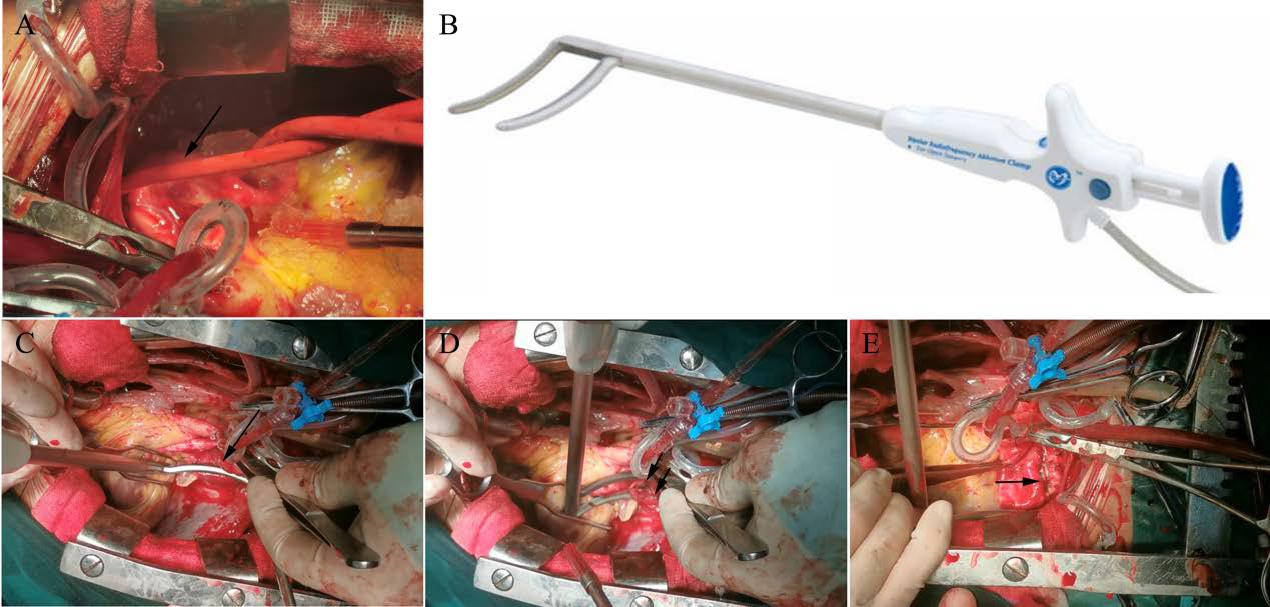
A straight vertical midline skin incision was made through a median sternotomy while placing the patient on cardiopulmonary bypass using bicaval cannulation. Initially, the space between the aorta and the dilated main pulmonary artery and the left pulmonary artery was bluntly dissected. Occasionally, a sharp division was necessary. After cardiopulmonary bypass, the posterior wall of the dilated main pulmonary artery, and the anterior and posterior walls of the right pulmonary artery were dissected. The bipolar clamp was placed from the right side, after carefully developing the space between the right pulmonary artery and the ascending aorta using blunt dissection to avoid any injury (Figure 1). On the left side, the ligament of Marshall was divided with cautery, while the left pulmonary artery was bluntly dissected and surrounded by a red catheter (Figure 2A).
Figure 1: Schematic illustration of pulmonary artery radiofrequency denervation.
PARFD was performed with linear ablation using bipolar radiofrequency (RF) energy simultaneously with cardiopulmonary bypass rewarming. The ablation device (Med-Zenith Ltd., Beijing, China) (Figure 2B) delivers RF energy through two closely approximated 6-cm electrodes embedded in the jaws of the clamp. The RF device was placed around the left pulmonary artery bifurcation (Figure 2C) for delivering the RF energy until the algorithm confirmed transmurality for two times (Figure 2D). Following the initial ablation, the device was unclamped, moved proximally by several millimeters on the left pulmonary artery, and re-clamped for a second ablation to ensure electrical isolation.
Following completion of the left pulmonary artery isolation, the dilated main pulmonary artery and the right pulmonary artery bifurcation were isolated in a similar manner with the RF device (Figure 2E).
Figure 2: The left pulmonary artery is bluntly dissected and surrounded with a red catheter (A). The radiofrequency ablation device (B). The bipolar radiofrequency device is placed around the left pulmonary artery bifurcation (C). The black arrows indicate the two ablation lines at the ostial left pulmonary artery (D). The black arrow indicates the two ablation lines at the bifurcation of the main PA (E).
Discussion
At first, we were planning to perform transthoracic denervation with the cut-and-sew method, or by stripping the adipose and connective tissues around the main pulmonary artery trunk and its bifurcation and the proximal regions of the left and right pulmonary artery, similar to the transthoracic PADN procedure in monocrotaline PH rat models [8] . However, these surgical procedures are time-consuming and it is challenging to achieve hemostasis. Considering the safety and efficacy of RF ablation in the treatment of atrial fibrillation, the bipolar RF ablation device may prove more effective and safer for transthoracic denervation.
Unlike the other techniques available for the treatment of PAH-CHD, such as sympathetic ganglion block, catheter-based renal denervation, and PADN, TPARFD is a novel therapeutic option, although an invasive and high-risk procedure. Therefore, it is better to perform PARFD only in adult PAH-CHD cases, concomitantly with cardiac operations.
In contrast to the percutaneous PADN, TPARFD is performed with two ablation lines at the ostial left pulmonary artery and the bifurcation of the main pulmonary artery and ostial right pulmonary artery, and not just the ten points embedded on the circular tip of the ablation catheter.
Conclusion
TPARFD is a novel and effective therapeutic option for the treatment of PAH-ACHD which may be performed concomitantly with other cardiac surgeries. Further clinical study is required to confirm the efficacy of TPARFD in PAH-ACHD.
Supplementary Materials
Not Applicable.
Declarations
Abbreviations
Not Applicable.
Author contributions
Not Applicable.
Acknowledgements
Not Applicable.
Funding information
This work was funded by the First Affiliated Hospital of Anhui Medical University Clinical Research Initiation Program (no. LCYJ2021YB016) and partly supported by the University Natural Science Research Project of Anhui Province (no. KJ2018A0180) .
Ethics approval and consent to participate
Not Applicable.
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
References
[1] Galie N, Manes A, Palazzini M, Negro L, Marinelli A, Gambetti S et al. Management of pulmonary arterial hypertension associated with congenital systemic-to-pulmonary shunts and Eisenmenger's syndrome. Drugs 2008;68:1049-66.
[2] van Dissel AC, Mulder BJ, Bouma BJ. The Changing Landscape of Pulmonary Arterial Hypertension in the Adult with Congenital Heart Disease. J Clin Med 2017;6:40.
[3] Boudjemline Y, Patel M, Malekzadeh-Milani S, Szezepanski I, Lévy M, Bonnet D. Patent ductus arteriosus stenting (transcatheter Potts shunt) for palliation of suprasystemic pulmonary arterial hypertension: a case series. Circ Cardiovasc Interv 2013;6:e18-20.
[4] Baruteau AE, Belli E, Boudjemline Y, Laux D, Lévy M, Simonneau G et al. Palliative Potts shunt for the treatment of children with drug-refractory pulmonary arterial hypertension: updated data from the first 24 patients. Eur J Cardiothorac Surg 2015;47:e105-10.
[5] Chen SL, Zhang FF, Xu J, Xie DJ, Zhou L, Nguyen T et al. Pulmonary artery denervation to treat pulmonary arterial hypertension: the single-center, prospective, first-in-man PADN-1 study (first-in-man pulmonary artery denervation for treatment of pulmonary artery hypertension). J Am Coll Cardiol 2013;62:1092-1100.
[6] Chen SL, Zhang H, Xie DJ, Zhang J, Zhou L, Rothman AM et al. Hemodynamic, functional, and clinical responses to pulmonary artery denervation in patients with pulmonary arterial hypertension of different causes: phase II results from the Pulmonary Artery Denervation-1 study. Circ Cardiovasc Interv 2015;8:e002837.
[7] Zhang H, Zhang J, Chen M, Xie DJ, Kan J, Yu W et al. Pulmonary Artery Denervation Significantly Increases 6-Min Walk Distance for Patients With Combined Pre- and Post-Capillary Pulmonary Hypertension Associated With Left Heart Failure: The PADN-5 Study. JACC Cardiovasc Interv 2019;12:274-84.
[8] Huang Y, Liu YW, Pan HZ, Zhang XL, Li J, Xiang L et al. Transthoracic Pulmonary Artery Denervation for Pulmonary Arterial Hypertension. Arterioscler Thromb Vasc Biol 2019;39:704-18.
 Figures
Figures References
References Peer
Peer Information
InformationFigure 1: Schematic illustration of pulmonary artery radiofrequency denervation.

Figure 2: The left pulmonary artery is bluntly dissected and surrounded with a red catheter (A). The radiofrequency ablation device (B). The bipolar radiofrequency device is placed around the left pulmonary artery bifurcation (C). The black arrows indicate the two ablation lines at the ostial left pulmonary artery (D). The black arrow indicates the two ablation lines at the bifurcation of the main PA (E).

[1] Galie N, Manes A, Palazzini M, Negro L, Marinelli A, Gambetti S et al. Management of pulmonary arterial hypertension associated with congenital systemic-to-pulmonary shunts and Eisenmenger's syndrome. Drugs 2008;68:1049-66.
[2] van Dissel AC, Mulder BJ, Bouma BJ. The Changing Landscape of Pulmonary Arterial Hypertension in the Adult with Congenital Heart Disease. J Clin Med 2017;6:40.
[3] Boudjemline Y, Patel M, Malekzadeh-Milani S, Szezepanski I, Lévy M, Bonnet D. Patent ductus arteriosus stenting (transcatheter Potts shunt) for palliation of suprasystemic pulmonary arterial hypertension: a case series. Circ Cardiovasc Interv 2013;6:e18-20.
[4] Baruteau AE, Belli E, Boudjemline Y, Laux D, Lévy M, Simonneau G et al. Palliative Potts shunt for the treatment of children with drug-refractory pulmonary arterial hypertension: updated data from the first 24 patients. Eur J Cardiothorac Surg 2015;47:e105-10.
[5] Chen SL, Zhang FF, Xu J, Xie DJ, Zhou L, Nguyen T et al. Pulmonary artery denervation to treat pulmonary arterial hypertension: the single-center, prospective, first-in-man PADN-1 study (first-in-man pulmonary artery denervation for treatment of pulmonary artery hypertension). J Am Coll Cardiol 2013;62:1092-1100.
[6] Chen SL, Zhang H, Xie DJ, Zhang J, Zhou L, Rothman AM et al. Hemodynamic, functional, and clinical responses to pulmonary artery denervation in patients with pulmonary arterial hypertension of different causes: phase II results from the Pulmonary Artery Denervation-1 study. Circ Cardiovasc Interv 2015;8:e002837.
[7] Zhang H, Zhang J, Chen M, Xie DJ, Kan J, Yu W et al. Pulmonary Artery Denervation Significantly Increases 6-Min Walk Distance for Patients With Combined Pre- and Post-Capillary Pulmonary Hypertension Associated With Left Heart Failure: The PADN-5 Study. JACC Cardiovasc Interv 2019;12:274-84.
[8] Huang Y, Liu YW, Pan HZ, Zhang XL, Li J, Xiang L et al. Transthoracic Pulmonary Artery Denervation for Pulmonary Arterial Hypertension. Arterioscler Thromb Vasc Biol 2019;39:704-18.
Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-03-05
Accepted 2025-05-27
Published 2025-06-30


