Abstract
Objective: To compare the characteristics of spontaneous nystagmus (SN) in patients with vestibular neuritis (VN) at different disease stages and to investigate its correlation with semicircular canal dysfunction.
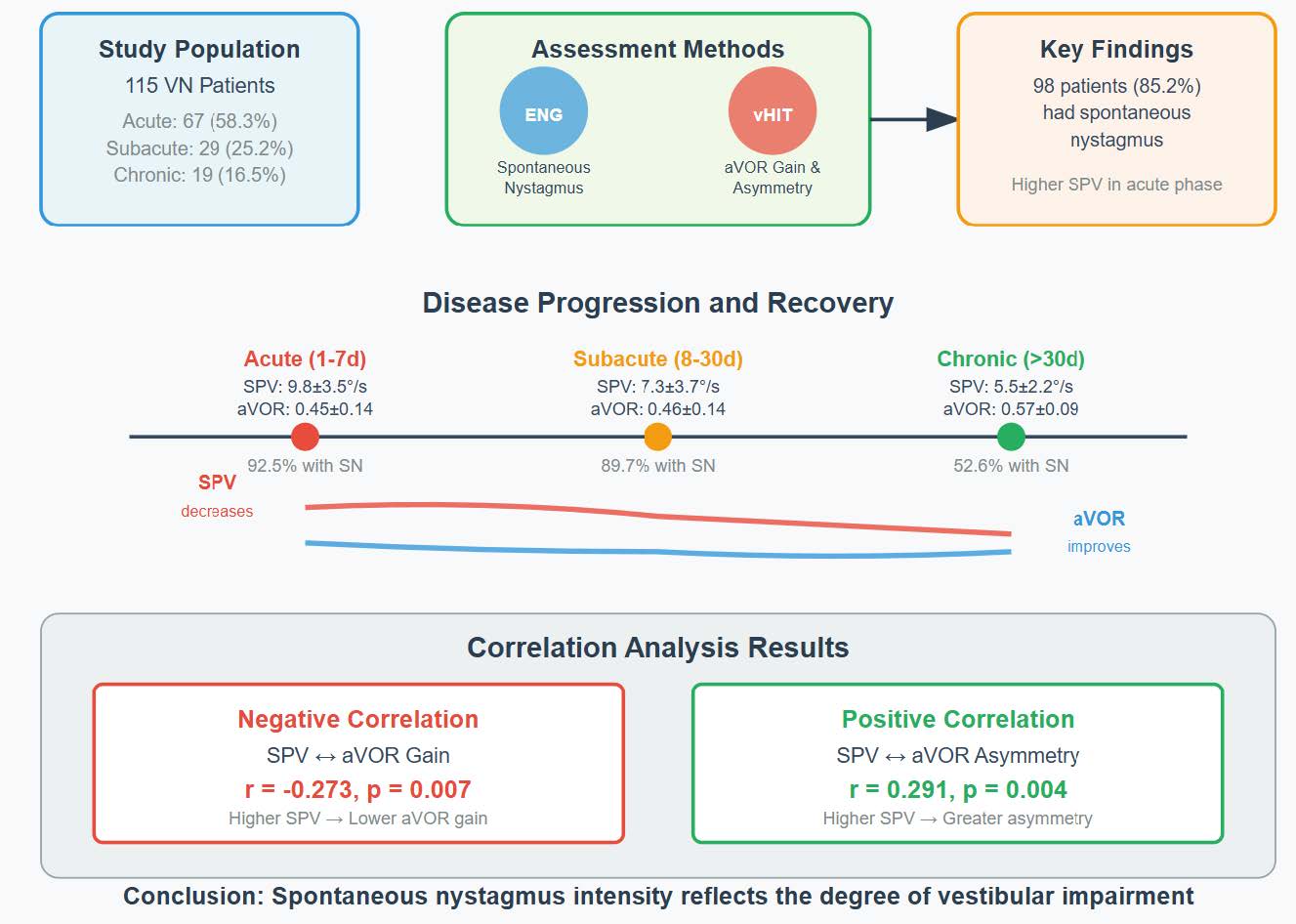
Methods: A total of 115 VN patients were enrolled, who were divided into acute phase (1 – 7 days), subacute phase (8 – 30 days) and chronic phase (>30 days) according to the course of disease. Quantitative assessment of SN was performed using electronystagmography (ENG), with particular focus on the slow phase velocity (SPV) in the horizontal plane. Simultaneously, vestibular-ocular reflex function was systematically evaluated through video head impulse test (vHIT), with quantitative measurements of angular vestibulo-ocular reflex (aVOR) gain and asymmetry of the affected semicircular canals.
Results: Among 115 patients, 67 (58.3%) were in acute phase, 29 (25.2%) in subacute phase and 19 (16.5%) in chronic phase. The mean age of patients in the acute phase was significantly lower than that in the chronic phase (p = 0.028), while there was no significant difference between the subacute phase and the other two groups. 98 patients (85.2%) had SN, 62 patients (92.5%) in acute phase, 26 patients (89.7%) in subacute phase and 10 patients (52.6%) in chronic phase. SPV in acute phase was significantly higher than that in subacute phase (p = 0.003) and chronic phase (p < 0.001), but there was no significant difference between chronic phase and subacute phase. In 112 patients with VN, the horizontal semicircular canals were affected, and the mean aVOR gain and asymmetry of the damaged side were 0.47 ± 0.14 and 0.35 ± 0.14, respectively. The aVOR gain of injured side in chronic phase was significantly higher than that in acute phase (p < 0.001) and subacute phase (p = 0.007), but there was no significant difference between acute phase and subacute phase (p = 0.950). The asymmetry in chronic phase was significantly lower than that in acute phase (p = 0.001) and subacute phase (p = 0.018), and there was no significant difference between acute phase and subacute phase (p = 0.312). SPV in horizontal component was negatively correlated with the aVOR gain of damaged horizontal semicircular canals (r = -0.273, p = 0.007), and positively correlated with aVOR asymmetry (r = 0.291, p = 0.004).
Conclusion: The existence of SN is helpful for diagnosis of VN, and its intensity can reflect the degree of vestibular impairment.
Keywords: vestibular neuritis; spontaneous nystagmus; video head impulse test
Introduction
Vestibular neuritis (VN) is an acute peripheral vestibular syndrome and the third most common cause of peripheral vestibular disorders. It is characterized by acute or, less frequently, subacute onset of rotational or non-rotational vertigo, accompanied by imbalance, nausea/vomiting, and/or oscillopsia [1] . The video head impulse test (vHIT) is a widely used tool for assessing semicircular canal function, capable of accurately localizing the affected canal and measuring the angular vestibulo-ocular reflex (aVOR) gain of all six semicircular canals [2] . In this study, we compared the extent of vestibular dysfunction and the slow phase velocity (SPV) in VN patients across different disease stages (acute, subacute, and chronic).
Methods
This is a single-center, retrospective study involving patients diagnosed with vestibular neuritis at the Headache and Vertigo Clinic of the Department of Neurology, The First Affiliated Hospital of Anhui Medical University, from June 2022 to May 2024.
Inclusion and Exclusion criteria
Inclusion Criteria: All patients underwent comprehensive neurological and otological examinations, as well as video infrared goggles testing. All patients also underwent both vHIT and electronystagmography (ENG). Patients with focal neurological signs underwent brain MRI. Patients with acute vestibular syndrome during vertigo attacks and vHIT results showing unilateral gain <0.7 were included in the subsequent analyses.
Exclusion Criteria: 1) Patients with central nervous system lesions confirmed by MRI. 2) Patients who had taken any vestibular suppressants within 48 hours before vestibular testing. 3) Patients who had undergone vestibular rehabilitation before ENG testing.
Study design
The vHIT device was used to examine the patients. Patients were seated with their heads flexed forward by 30° to align the horizontal semicircular canals parallel to the horizontal plane. They were instructed to fixate on a stationary target 1 m ahead. The examiner then abruptly and randomly rotated the patient's head to the left and right (head rotation angle of 20° – 30°, lasting 150 – 200 ms, with a peak head velocity >150°/s). Subsequently, the patient's head was turned 45° to the left and right, and the examiner performed abrupt, small-amplitude, random up-and-down head rotations in the sagittal plane. Each direction was tested with 20 effective head impulses. The goggles recorded the eye and head movement trajectories and saccades during head rotation. The aVOR gain for each head impulse was calculated as the ratio of peak eye velocity to peak head velocity, and the average value was taken as the gain for each semicircular canal.
During the ENG examination, the presence of SN in the neutral head position and while looking straight ahead was assessed. Instruct the patient to sit upright, wear the goggles, eliminate visual fixation, and record nystagmus for at least 30 seconds. For patients with SN, the horizontal component of the SPV was recorded. Patients were categorized into three groups based on the time interval from symptom onset to examination: acute phase (1 – 7 days), subacute phase (8 – 30 days), and chronic phase (>30 days). We compared the differences in age, gender, aVOR gain and asymmetry among the three groups. The aVOR asymmetry was calculated using the relative asymmetry formula: (higher mean high-speed aVOR gain - lower mean high-speed aVOR gain)/ (higher mean high-speed aVOR gain + lower mean high-speed aVOR gain).
Statistic analysis
Data were analyzed using SPSS version 26, and figures were created using GraphPad Prism version 9.5. Normally distributed data are presented as mean ± standard deviation, while non-normally distributed data are presented as median and interquartile range. For comparisons of continuous variables between groups, normally distributed data were analyzed using independent samples t-tests, while non-normally distributed data were analyzed using the Mann-Whitney U test (a type of rank-sum test). Categorical data were analyzed using Pearson's chi-square test or Fisher's exact test, as appropriate. All results were considered statistically significant with a two-sided p-value < 0.05.
Results
A total of 115 patients were included in this study, of whom 74 were male (64.3%), with a mean age of 50.2 ± 14.4 years. Vestibular dysfunction was observed on the left side in 75 patients (65.2%) and on the right side in 40 patients (34.8%). The superior vestibular nerve was affected in 108 cases (93.9%), the inferior vestibular nerve in 2 cases (1.7%), and both nerves in 5 cases (4.3%). The horizontal semicircular canal was affected in 112 patients (97.4%), the anterior semicircular canal in 17 patients (14.8%), and the posterior semicircular canal in 7 patients (6.1%). The acute phase was observed in 67 patients (58.3%), the subacute phase in 29 patients (25.2%), and the chronic phase in 19 patients (16.5%). The mean age of patients in the acute phase was 48.3 ± 15.2 years, which was significantly lower than that of the chronic phase (56.6 ± 13.3 years, p = 0.028), while the subacute phase (50.3 ± 12.5 years) showed no significant difference compared with the other two groups. There was no significant difference in the male-to-female ratio among the three groups (p = 0.112).
Spontaneous nystagmus
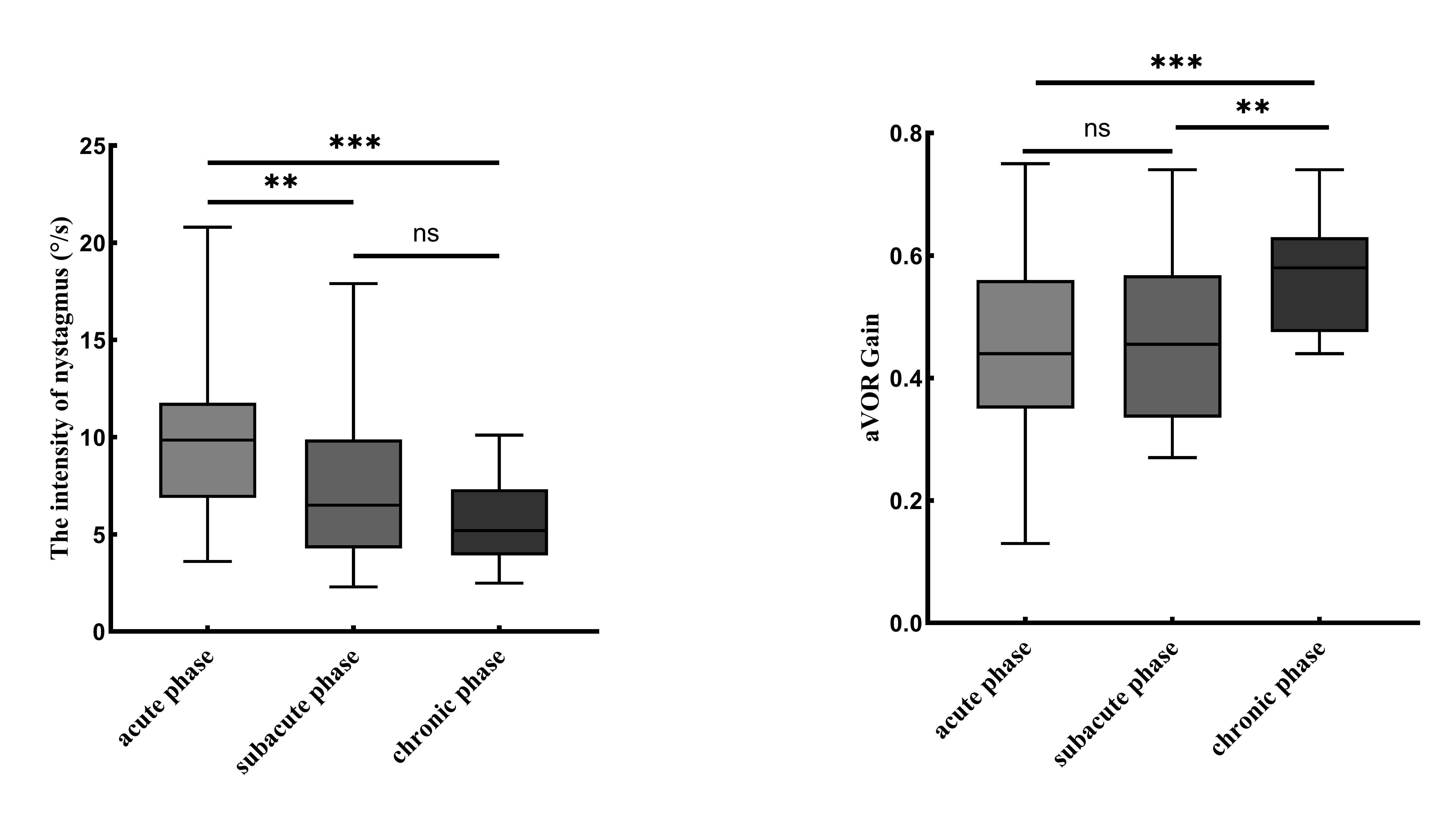
SN was observed in 98 patients (85.2%). Among them, 62 patients (92.5%) were in the acute phase, 26 (89.7%) in the subacute phase, and 10 (52.6%) in the chronic phase. Of these patients with SN, 95 (96.9%) exhibited a fast phase directed toward the healthy side, while 3 (3.1%) had a fast phase directed toward the affected side. The mean SPV of horizontal nystagmus was 8.7 ± 3.8°/s. The SPV in the acute phase (9.8 ± 3.5°/s) was significantly higher than that in the subacute phase (7.3 ± 3.7°/s, p = 0.003) and the chronic phase (5.5 ± 2.2°/s, p < 0.001), while no significant difference was observed between the subacute and chronic phases (p = 0.173) (Fig. 1).
Figure 1. Comparison of SN and lateral semicircular canal aVOR gain in VN patients with different disease courses (acute, subacute and chronic), **p ≤ 0.01, **p ≤ 0.001.
vHIT Results
Among the 112 patients with VN involving the horizontal semicircular canal, the mean gain value of the affected side was 0.47 ± 0.14, and the mean asymmetry ratio was 0.35 ± 0.14. These patients were distributed across three phases: acute phase (67 cases), subacute phase (28 cases), and chronic phase (17 cases). The aVOR gain of the affected side in the chronic phase (0.57 ± 0.09) was significantly higher than that in the acute phase (0.45 ± 0.14, p < 0.001) and subacute phase (0.46 ± 0.14, p = 0.007), while no significant difference was observed between the acute and subacute phases (p = 0.950). The aVOR asymmetry in the chronic phase (0.25 ± 0.09) was significantly lower than that in the acute phase (0.38 ± 0.14, p = 0.001) and subacute phase (0.35 ± 0.14, p = 0.018), with no significant difference between the acute and subacute phases (p = 0.312) (Fig. 1). In the study, 17 patients had involvement of the anterior semicircular canal, with 10 cases in the acute phase, 5 in the subacute phase, and 2 in the chronic phase. The mean aVOR gain of the affected side was 0.55 ± 0.09, and the mean aVOR asymmetry was 0.25 ± 0.11. Additionally, 7 patients had involvement of the posterior semicircular canal, with 2 cases in the acute phase, 3 in the subacute phase, and 2 in the chronic phase. The mean aVOR gain of the affected side was 0.57 ± 0.10, and the mean aVOR asymmetry was 0.24 ± 0.09.
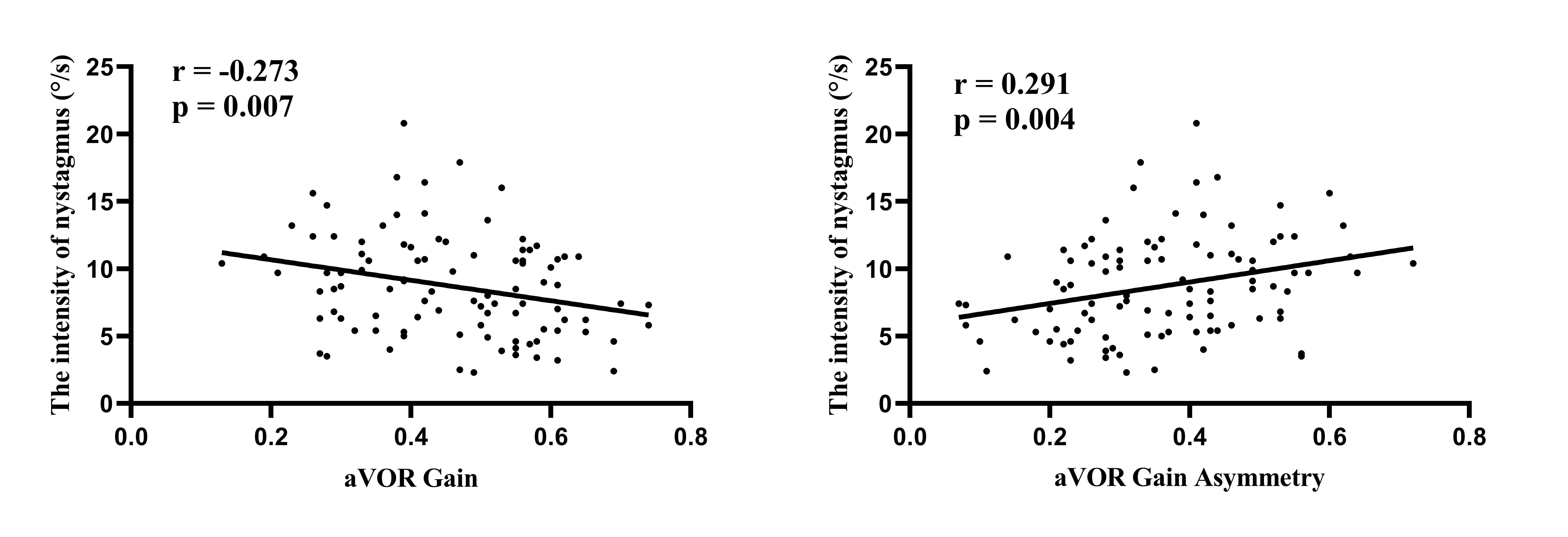
Among the 98 patients with SN, all exhibited involvement of the horizontal semicircular canal. The SPV of the horizontal component of nystagmus was significantly negatively correlated with the aVOR gain of the affected horizontal semicircular canal (r = -0.273, p = 0.007) and significantly positively correlated with the aVOR asymmetry (r = 0.291, p = 0.004) (Fig. 2).
Figure 2. Correlation analysis of SN on horizontal component with gain value and asymmetry ratio of damaged side in VN patients.
Discussion
The results of this study indicate that involvement of the superior vestibular nerve is the most common (93.9%) among patients with VN, followed by involvement of both the superior and inferior vestibular nerves (4.3%), while isolated involvement of the inferior vestibular nerve is the rarest (1.7%). These findings are consistent with those reported in previous literature [1] . These findings are related to the anatomical characteristics of the vestibular system. First, the osseous canal through which the superior vestibular nerve travels is relatively long and narrow, making it more susceptible to compression when the nerve becomes swollen. Second, latent infections of herpes simplex virus type 1 (HSV-1) in the vestibular ganglion are predominantly located in the superior vestibular ganglion, rendering the superior vestibular nerve more vulnerable to HSV-1 infection [3] .
In this study, we found that patients with chronic VN had significantly higher ages compared to the other two groups, suggesting that older patients are more likely to experience chronicity of symptoms. Previous studies have reported that in the follow-up of VN patients, the degree of symptom improvement in the elderly group was significantly lower than that in the younger group [4,5] . This phenomenon may be attributed to the multisystem degeneration in elderly patients, including the degeneration of the vestibular and visual systems. Therefore, for elderly patients, it is particularly important to develop an early and tailored vestibular rehabilitation therapy (VRT) plan. In addition to vestibular exercises, vision plays a crucial role in compensating for vestibular deficits in older adults. For elderly patients with blurred vision, such as those with cataracts, vision correction should be implemented to prevent imbalance and falls.
After vestibular dysfunction, clinical symptoms can be categorized into static symptoms, which occur in a resting state, and dynamic symptoms, which emerge during body movement. Static symptoms include SN, contrapositional deviation, ocular torsion, head and body tilt, vertigo, and autonomic nervous system responses such as nausea and vomiting. Dynamic symptoms involve changes in aVOR gain, among others. Following vestibular dysfunction, the body gradually alleviates symptoms through vestibular compensation, which is divided into static and dynamic compensation. Static compensation is a transient, spontaneous, neurophysiological change based on the vestibular nuclear complex, while dynamic compensation is a long-term, learned process that involves multiple central nervous system mechanisms. It has been reported in the literature that rodents can complete static vestibular compensation within one week [6] . It has been reported in the literature that rodents can complete static vestibular compensation within one week [7] , while cats can complete this process within six weeks [8,9] . Early studies by Matsuzaki et al. have shown that SN typically disappears completely within 1 month after the onset of vestibular neuritis [10] . In this study, we found that after removing fixation suppression in the dark condition, more than 50% of patients with chronic vestibular neuritis still exhibited SN, which can serve as an important clinical indicator for the diagnosis of the chronic phase of vestibular neuritis. In our study, the SPV in the acute phase was significantly higher than that in the subacute and chronic phases, with no significant difference between the subacute and chronic phases. Additionally, the aVOR gain on the affected side in the chronic phase was significantly higher than that in the acute and subacute phases, while the aVOR asymmetry was significantly lower than that in the acute and subacute phases; there were no significant differences between the acute and subacute phases. These results suggest that static compensation occurs early in the disease course, while dynamic compensation requires a longer period of time.
After vestibular nerve damage, the generation of SN is due to the reduced resting activity on the affected side, resulting in a neurophysiological imbalance between the bilateral vestibular nuclei [11] . Studies have shown that in the early stages of the disease, the slow phase of SN is directed toward the affected side. As vestibular function recovers on the affected side and static compensation occurs, the SPV gradually diminishes and eventually disappears. In long-term follow-up studies of patients with unilateral vestibular dysfunction, the duration of SN shows significant individual variability, ranging from several days to months or even years. This variability is primarily determined by the rate of vestibular compensation and differences in individual recovery capabilities [12,13] . In the patients included in this study, vestibular function remained impaired, with over 50% of patients in the chronic phase still exhibiting SN. This suggests that static compensation in these patients is insufficient to overcome the imbalance between the bilateral vestibular systems. Our study found that the SPV is negatively correlated with the aVOR gain on the affected side and positively correlated with the aVOR asymmetry of bilateral gain values. These findings are consistent with previous studies [14] . These findings indicate that the presence of SN not only suggests an imbalance in bilateral vestibular function but also reflects the degree of vestibular dysfunction based on its intensity.
Conclusion
In summary, among patients with VN, older patients tend to experience longer-lasting symptoms and would benefit from early implementation of tailored rehabilitation programs. The persistence of SN in the chronic phase serves as an important diagnostic indicator and reflects the severity of vestibular dysfunction based on its intensity. As a cross-sectional study, our research has the following limitations: First, the study design does not allow for the assessment of the dynamic evolution of vestibular dysfunction over time. Future research should involve longitudinal cohort studies with long-term follow-up of at least 6–12 months to elucidate the temporal characteristics of vestibular compensation. Second, the current evaluation primarily relies on objective measures of vestibular function. We recommend incorporating subjective assessment tools, such as the Vestibular Symptom Scale and the Dizziness Handicap Inventory, to comprehensively evaluate the extent of vestibular dysfunction and its impact on quality of life.
Acknowledgements
The authors would like to thank all the patients.
Author contributions
JYW and JC conceived and designed the study, and completed the first draft. JC acquired the data. JYW analyzed the data. All authors critically review the content of the manuscript. All authors approved the final version.
Ethics approval and consent to participate
The study protocols were approved by the Institutional Review Board of the First Affiliated Hospital of Anhui Medical University (no. PJ2024-10-65).
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
References
[1] Strupp M, Bisdorff A, Furman J, Hornibrook J, Jahn K, Maire R, Newman-Toker D, Magnusson [2] (2022) Acute unilateral vestibulopathy/vestibular neuritis: Diagnostic criteria. Journal of vestibular research : equilibrium & orientation 32(5):389-406. https:// doi.org/10.3233/ves-220201
[3] Jasinska-Nowacka A, Niemczyk K (2024) Application of a Video Head Impulse Test in the Diagnosis of Vestibular Neuritis. Life (Basel) 14(6). https:// doi.org/10.3390/life14060757
[4] Gianoli G, Goebel J, Mowry S, Poomipannit P (2005) Anatomic differences in the lateral vestibular nerve channels and their implications in vestibular neuritis. Otol Neurotol 26(3):489-494. https:// doi.org/10.1097/01.mao.0000169787.99835.9f
[5] Jacobson G, McCaslin D (2020) Vestibular Neuritis in Patients Among Different Age Groups: Clinical Features and Outcomes. J Am Acad Audiol 31(9):627-628. https:// doi.org/10.1055/s-0041-1723789
[6] Scheltinga A, Honegger F, Timmermans DP, Allum JH (2016) The Effect of Age on Improvements in Vestibulo-Ocular Reflexes and Balance Control after Acute Unilateral Peripheral Vestibular Loss. Front Neurol 7:18. https:// doi.org/10.3389/fneur.2016.00018
[7] Lacour M, Helmchen C, Vidal PP (2016) Vestibular compensation: the neuro-otologist's best friend. J Neurol 263 Suppl 1:S54-64. https:// doi.org/10.1007/s00415-015-7903-4
[1] Darlington CL, Smith PF (2000) Molecular mechanisms of recovery from vestibular damage in mammals: recent advances. Prog Neurobiol 62(3):313-325. https:// doi.org/10.1016/s0301-0082(00)00002-2
[8] Lacour M, Tighilet B (2010) Plastic events in the vestibular nuclei during vestibular compensation: the brain orchestration of a "deafferentation" code. Restor Neurol Neurosci 28(1):19-35. https:// doi.org/10.3233/rnn-2010-0509
[9] Borel L, Lopez C, Péruch P, Lacour M (2008) Vestibular syndrome: a change in internal spatial representation. Neurophysiol Clin 38(6):375-389. https:// doi.org/10.1016/j.neucli.2008.09.002
[10] Matsuzaki M, Kamei T (1995) Stage-assessment of the progress of continuous vertigo of peripheral origin by means of spontaneous and head-shaking nystagmus findings. Acta Otolaryngol Suppl 519:188-190. https:// doi.org/10.3109/00016489509121900
[11] Robinson DA, Zee DS, Hain TC, Holmes A, Rosenberg LF (1984) Alexander's law: its behavior and origin in the human vestibulo-ocular reflex. Ann Neurol 16(6):714-722. https:// doi.org/10.1002/ana.410160614
[12] Choi KD, Oh SY, Kim HJ, Koo JW, Cho BM, Kim JS (2007) Recovery of vestibular imbalances after vestibular neuritis. The Laryngoscope 117(7):1307-1312. https:// doi.org/10.1097/MLG.0b013e31805c08ac
[13] Bergenius J, Perols O (1999) Vestibular neuritis: a follow-up study. Acta Otolaryngol 119(8):895-899. https:// doi.org/10.1080/00016489950180243
[14] Zhang X, Deng Q, Liu Y, Li S, Wen C, Liu Q, Huang X, Wang W, Chen T (2023) Characteristics of spontaneous nystagmus and its correlation to video head impulse test findings in vestibular neuritis. Front Neurosci 17:1243720. https:// doi.org/10.3389/fnins.2023.1243720
 Figures
Figures References
References Peer
Peer Information
InformationFigure 1. Comparison of SN and lateral semicircular canal aVOR gain in VN patients with different disease courses (acute, subacute and chronic), **p ≤ 0.01, **p ≤ 0.001.

Figure 2. Correlation analysis of SN on horizontal component with gain value and asymmetry ratio of damaged side in VN patients.

[1] Strupp M, Bisdorff A, Furman J, Hornibrook J, Jahn K, Maire R, Newman-Toker D, Magnusson [2] (2022) Acute unilateral vestibulopathy/vestibular neuritis: Diagnostic criteria. Journal of vestibular research : equilibrium & orientation 32(5):389-406. https:// doi.org/10.3233/ves-220201
[3] Jasinska-Nowacka A, Niemczyk K (2024) Application of a Video Head Impulse Test in the Diagnosis of Vestibular Neuritis. Life (Basel) 14(6). https:// doi.org/10.3390/life14060757
[4] Gianoli G, Goebel J, Mowry S, Poomipannit P (2005) Anatomic differences in the lateral vestibular nerve channels and their implications in vestibular neuritis. Otol Neurotol 26(3):489-494. https:// doi.org/10.1097/01.mao.0000169787.99835.9f
[5] Jacobson G, McCaslin D (2020) Vestibular Neuritis in Patients Among Different Age Groups: Clinical Features and Outcomes. J Am Acad Audiol 31(9):627-628. https:// doi.org/10.1055/s-0041-1723789
[6] Scheltinga A, Honegger F, Timmermans DP, Allum JH (2016) The Effect of Age on Improvements in Vestibulo-Ocular Reflexes and Balance Control after Acute Unilateral Peripheral Vestibular Loss. Front Neurol 7:18. https:// doi.org/10.3389/fneur.2016.00018
[7] Lacour M, Helmchen C, Vidal PP (2016) Vestibular compensation: the neuro-otologist's best friend. J Neurol 263 Suppl 1:S54-64. https:// doi.org/10.1007/s00415-015-7903-4
[1] Darlington CL, Smith PF (2000) Molecular mechanisms of recovery from vestibular damage in mammals: recent advances. Prog Neurobiol 62(3):313-325. https:// doi.org/10.1016/s0301-0082(00)00002-2
[8] Lacour M, Tighilet B (2010) Plastic events in the vestibular nuclei during vestibular compensation: the brain orchestration of a "deafferentation" code. Restor Neurol Neurosci 28(1):19-35. https:// doi.org/10.3233/rnn-2010-0509
[9] Borel L, Lopez C, Péruch P, Lacour M (2008) Vestibular syndrome: a change in internal spatial representation. Neurophysiol Clin 38(6):375-389. https:// doi.org/10.1016/j.neucli.2008.09.002
[10] Matsuzaki M, Kamei T (1995) Stage-assessment of the progress of continuous vertigo of peripheral origin by means of spontaneous and head-shaking nystagmus findings. Acta Otolaryngol Suppl 519:188-190. https:// doi.org/10.3109/00016489509121900
[11] Robinson DA, Zee DS, Hain TC, Holmes A, Rosenberg LF (1984) Alexander's law: its behavior and origin in the human vestibulo-ocular reflex. Ann Neurol 16(6):714-722. https:// doi.org/10.1002/ana.410160614
[12] Choi KD, Oh SY, Kim HJ, Koo JW, Cho BM, Kim JS (2007) Recovery of vestibular imbalances after vestibular neuritis. The Laryngoscope 117(7):1307-1312. https:// doi.org/10.1097/MLG.0b013e31805c08ac
[13] Bergenius J, Perols O (1999) Vestibular neuritis: a follow-up study. Acta Otolaryngol 119(8):895-899. https:// doi.org/10.1080/00016489950180243
[14] Zhang X, Deng Q, Liu Y, Li S, Wen C, Liu Q, Huang X, Wang W, Chen T (2023) Characteristics of spontaneous nystagmus and its correlation to video head impulse test findings in vestibular neuritis. Front Neurosci 17:1243720. https:// doi.org/10.3389/fnins.2023.1243720
Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-01-30
Accepted 2025-03-22
Published 2025-03-30


