Abstract
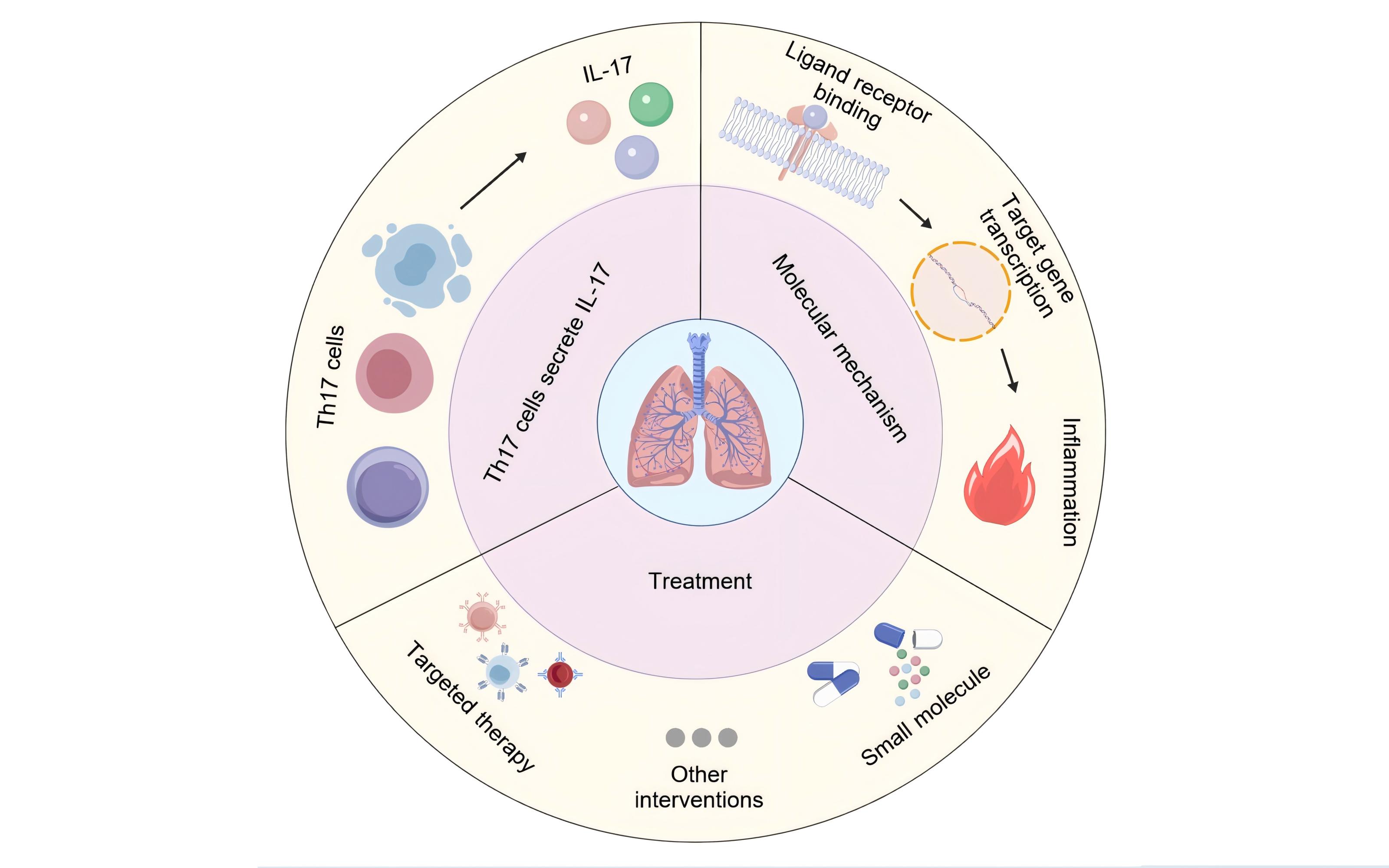
Pulmonary fibrosis is a serious lung disease characterized by the destruction of alveolar structures and excessive proliferation of fibrous tissue. The interleukin-17 (IL-17) family consists of six members (IL-17A-17F), which play a crucial role in the occurrence and development of pulmonary fibrosis. The IL-17 family drives pulmonary fibrosis through multiple mechanisms such as pro-inflammatory cytokines, immune cell recruitment, and fibroblast activation. IL-17A is the core molecule, while other members participate in the disease process through synergistic or independent pathways. Targeting the IL-17 signaling axis provides a new strategy for the treatment of pulmonary fibrosis. This article summarizes the effects of IL-17 on pulmonary inflammation response and fibrosis process through literature review, as well as its possible involvement in signal transduction molecular mechanisms, and explores its potential as a therapeutic target, providing theoretical basis for future research aimed at regulating IL-17 expression and function to treat related diseases.
Keywords:interleukin-17 (IL-17) family, IL-17 receptor, pulmonary fibrosis
Introduction
Pulmonary fibrosis is a chronic progressive lung disease characterized by fibrosis of lung tissue and scar formation. This disease can damage the alveolar structure, impair gas exchange function, and ultimately lead to respiratory failure [1] . From the perspective of histopathology, this disease presents obvious heterogeneity characteristics, specifically manifested as thickening of alveolar walls, infiltration of inflammatory cells, and abnormal proliferation of fibroblasts accompanied by a large amount of extracellular matrix deposition. Interstitial lung disease (ILD) and idiopathic pulmonary fibrosis (IPF) are the most severe and irreversible types of progressive pulmonary fibrosis [2] .
In 2014, the US Food and Drug Administration (FDA) approved two drugs, pirfenidone and nintedanib, for the treatment of pulmonary fibrosis [3] . However, there is currently a lack of effective treatment options for pulmonary fibrosis, and available treatment methods can only delay the progression of the disease and cannot completely cure it. Therefore, a deeper understanding of the pathogenesis of pulmonary fibrosis and the search for new therapeutic targets have important clinical significance.
IL-17 is a cytokine secreted by Th17 cells and was initially found to be involved in the pathological processes of autoimmune and inflammatory diseases. The latest research confirms that IL-17 plays a crucial role in the occurrence and development of pulmonary fibrosis. This article will review the role of IL-17 in pulmonary fibrosis, providing reference for further research and treatment of pulmonary fibrosis.
IL-17 cytokine family
Overview
The IL-17 family consists of six structurally related cytokine members: IL-17A, IL-17B, IL-17C, IL-17D, IL-17E (IL-25), and IL-17F [4] . IL-17A is the first family member discovered and the most extensively studied cytokine. Researchers cloned the molecule using T-cell hybridoma technology in 1993 [5] , and officially named it cytotoxic T lymphocyte associated antigen 8 (CTLA-8). IL-17B and IL-17C were identified through sequence similarity alignment in the expression sequence tag database [6] . IL-17D, IL-17E, and IL-17F were mainly discovered through sequence homology search of genomic DNA sequences.
The molecular weight distribution of members in this family is between 30-52 kDa, and they have overlapping but different biological activities. IL-17A is a homodimeric glycoprotein composed of 155 amino acids linked by disulfide bonds. Its molecular weight with IL-17E is approximately 30 kDA, while IL-17B, IL-17C, and IL-17F are approximately 40 kDA. IL-17D has the highest molecular weight at 52 kDA [7] . Except for IL-17B, all other members form homodimeric structures through disulfide bonds. Sequence analysis shows that IL-17F is most similar to IL-17A, with 55% sequence homology, and they often co express. The homology between IL-17B, IL-17C, and IL-17D is 23-29%, while the homology between IL-17E and IL-17A is only 17%, making it the most diverse subtype in the family (Table 1) [8] .
Table 1. The molecular weight and sequence homology of IL-17 family members
|
IL-17 Subtype |
Size (kDA) |
Sequence homology with IL-17A(%) |
|
A |
35 |
- |
|
B |
41 |
29 |
|
C |
40 |
23 |
|
D |
52 |
25 |
|
E |
34 |
17 |
|
F |
44 |
55 |
IL-17 cytokine family receptors
The IL-17 receptor (IL-17R) is a type I transmembrane protein consisting of an extracellular domain of 293 amino acids, a transmembrane domain of 21 amino acids, and a cytoplasmic tail of 525 amino acids. The IL-17 receptor family consists of five subunits, namely IL-17RA, IL-17RB, IL-17RC, IL-17RD, and IL-17RE, with IL-17RA being a common receptor [9] . IL-17A and IL-17F bind to the dimer IL17RA/RC complex, IL-17B and IL-17E bind to the dimer 17RA/RB complex, and IL-17C binds to the IL-17RA/RE complex. However, the subunits of IL-17RD specific heterodimers have not yet been determined [10] . IL-17 participates in chronic and persistent inflammation, autoimmunity, and maintaining epithelial integrity by binding to the IL-17 receptor. These receptors share a unique protein-protein interaction domain at their cytoplasmic tails, called the SEF/IL-17R (SEFIR) domain. NF-κB activator 1 (Act1) is an activator of NF-κB and a member of the SEFIR protein family. It is also a key component of IL-17 signaling transduction [11] . The IL-17 receptor binds to IL-17 family cytokines in the form of homodimers or heterodimers, and transmits signals through adaptor protein 1 (ACT1) and E3 ubiquitin ligase tumor necrosis factor receptor associated factor protein 6 (TRAF6), leading to activation of nuclear factor kappa B (NF-κB), mitogen activated protein kinase (MAPK), and CCAAT/enhancer binding protein (C/EBP) pathways [12] . The signal transduction mediated by IL-17-Act1 leads to pro-inflammatory and neutrophil mobilization of cytokines and chemokines, including chemokine (C-X-C Motif) ligand- 1 (CXCL1), tumor necrosis factor (TNF), IL-6, and colony stimulating fctor 2 (CSF2) [13] . The IL-17A receptor is also commonly expressed in non hematopoietic cells, including epithelial cells and fibroblasts [14] . They play a crucial role in the development of pulmonary fibrosis and differentiation into myofibroblasts in EMT, leading to enhanced extracellular matrix deposition.
Cell sources of IL-17 cytokine family
Although Th17 cells are generally considered the main source of IL-17, this cytokine is also produced by gamma delta T cells, cytotoxic T cells (CD8+alpha beta T cells), natural killer (NK) cells, invariant natural killer T cells (iNKT), innate lymphocytes (ILC), neutrophils, eosinophils, and macrophages, known as type 17 cells [15] . IL-17A can be secreted by various cell types, including Th17CD8+T (Tc17), γδ T17, Innate immunity and non hematopoietic cells. IL-17B originates from neutrophils, chondrocytes, neurons, stromal cells, intestinal epithelial cells and B cells (germinal and memory cells). IL-17C originates from epithelial cells. IL-17D comes from fibroblasts, colonic epithelial cells, brain, skeletal muscle, adipose tissue, heart, lungs and pancreas. IL-17E is produced by mast cells, alveolar macrophages, eosinophils, basophils, ILC2, dendritic cells, stromal cells, epithelial cells and Th2 cells secrete. IL-17F is composed of Th17, Lymphoid tissue inducer, NK, iNKT, Neutrophils, ILC3 and gamma delta T cells secrete (Table 2) [16] .
Table 2. Cell sources of IL-17 cytokine family
|
IL-17 Subtype |
Cell sources |
|
A |
Th17CD8+T(Tc17), γδ T17, Innate immunity and non hematopoietic cells |
|
B |
neutrophils, chondrocytes, neurons, stromal cells, intestinal epithelial cells and B cells (germinal and memory cells) |
|
C |
epithelial cells |
|
D |
fibroblasts, colonic epithelial cells, brain, skeletal muscle, adipose tissue, heart, lungs and pancreas |
|
E |
mast cells, alveolar macrophages, eosinophils, basophils, ILC2, dendritic cells, stromal cells, epithelial cells and Th2 cells |
|
F |
Th17, Lymphoid tissue inducer, NK, iNKT, Neutrophils, ILC3 and gamma delta T cells |
Note: ILC2 type 2 innate lymphoid cells; NK natural killer; iNKT invariant natural killer T cells; ILC3 type 3 innate lymphoid cells
The role of IL-17 in promoting inflammation and pulmonary fibrosis
Acute Inflammation: Its Role in Pneumonia
Inflammation is the early stage of fibrosis. IL-17A participates in fibrosis development by regulating early pulmonary inflammation. In the pulmonary injury model induced by bleomycin (BLM), IL-17A expression is significantly upregulated, stimulating endothelial and epithelial cells to secrete pro-inflammatory factors such as Tumor Necrosis Factor alpha (TNF - α), IL-1, IL-6, Transforming Growth Factor beta (TGF - β), while promoting the production of chemokines such as IL-8, CCL1, CXCL2, and CXCL5. These molecules mediate the recruitment of inflammatory cells to the alveolar surface, driving the inflammatory cascade and accelerating the progression of pulmonary fibrosis [17] . Multiple studies have shown that BLM stimulation triggers neutrophil infiltration through the IL-17-dependent pathway, significantly increasing IL-6 and IL-1 β levels, ultimately promoting abnormal collagen deposition [18] . Therefore, intraperitoneal anti-IL-17A blockade of IL-17A can alleviate acute inflammation and fibrosis characteristics in mice [19] .
It is worth noting that clearing alveolar macrophages can downregulate the expression of IL-23 and IL-1 β, inhibit IL-17 activity, and alleviate early pulmonary inflammation and fibrosis caused by silica exposure [20] . In the IL-17 family, IL-17F has the highest sequence homology (55%) with IL-17A. It plays a key role in chronic inflammatory lung disease by inducing the production of IL-6 and chemokines in human tracheal epithelial cells, venous endothelial cells, and fibroblasts [21] . Further research has shown that IL-17C can stimulate Th17 cells to secrete a large amount of IL-17A, IL-17F, and IL-22, indicating its ability to enhance the effector function of Th17 cells. High expression of IL-17C in inflammatory tissues can exacerbate local inflammatory reactions [22] . The functional research of IL-17B is not yet sufficient, but existing evidence shows that some of its functions overlap with IL-17A and have pro-inflammatory properties under specific conditions, such as inducing peritoneal exudate cells and 3T3 cell lines to produce IL-6, IL-23, and IL-1 α, and promoting pulmonary fibrosis by synergistically stimulating Th17 cells to secrete neutrophil chemokines through TNF-α stimulation [23] . At present, the understanding of IL-17D is still limited. This molecule is mainly detected in activated B cells and resting CD4+T cells, and it participates in immune regulation by promoting the secretion of pro-inflammatory cytokines by endothelial cells [24] .
Chronic fibrosis: role in pulmonary fibrosis
Idiopathic pulmonary fibrosis is a progressive pulmonary interstitial fibrosis of unknown etiology, typically fatal and associated with chronic inflammation and tissue repair dysfunction.
Research has revealed that the IL-17 signaling pathway regulated by IL-1 β and IL-23 plays a critical role in the early stages of disease. This pathway activates neutrophil inflammatory response and upregulates fibroblast matrix metalloproteinase expression, driving the progression of pulmonary fibrosis [25] . Clinical data shows that elevated neutrophil count in bronchoalveolar lavage fluid of IPF patients can serve as a predictive indicator of early mortality risk [26] .
Further research has found that Th17 cells in IPF patients secrete abnormally high concentrations of TGF-β and IL-17A. In the mouse pulmonary fibrosis model induced by bleomycin, these cytokines significantly increased collagen deposition. The co culture experiment of human lung fibroblasts and Th17 cells also confirmed that it promotes the excessive generation of extracellular matrix components. It is worth noting that the pro fibrotic effect of IL-17 has cross disease commonalities: studies have shown that IL-17 is highly expressed in the bronchial mucosa of severe asthma patients and induces epithelial mesenchymal transition (EMT) in human small airway epithelial cells in vitro [27] . In addition, IL-17 mediates the EMT process of salivary gland epithelial cells in healthy individuals through an IL-22 dependent mechanism, revealing its important role in the progression of Sjogren's syndrome fibrosis [28] .
IL-17A
The study of experimental animal models has confirmed the crucial role of IL-17A in regulating the complex interactions between lung inflammation and fibrosis. In the bleomycin induced pulmonary fibrosis model, the expression level of IL-17A was significantly increased. IL-17A exacerbates the pathological process of pulmonary fibrosis by activating inflammatory responses and promoting collagen production [29] . Experiments have shown that IL-17A receptors are widely expressed on the cell membrane surfaces of lung epithelial cells and fibroblasts, which are involved in the pathological process of epithelial mesenchymal transition associated with pulmonary fibrosis. In addition, these cells also regulate the transformation of fibroblasts into myofibroblasts and increase the deposition of extracellular matrix [30] . It is worth noting that the IL-17A co factor IL-22 exhibits antagonistic effects: It exerts anti-fibrotic effects by inhibiting the EMT process in the bleomycin model [31] .
In addition, IL-17A promotes fibrosis by inhibiting autophagy activity in epithelial cells, while neutralizing IL-17A can enhance autophagy levels and accelerate collagen degradation in lung tissue [32] . In some studies, upregulation of IL-17R expression was detected in fibroblasts after treatment with bleomycin. The addition of exogenous IL-17A can promote the proliferation of fibroblasts and specifically induce the synthesis of α -smooth muscle actin (α-SMA) and collagen [33] . This process relies on signal transduction mediated by NF-κ B activator 1 (Act1): Act1, as a key adaptor protein for IL-17 receptor signaling, regulates fibrosis process by activating the NF- κ B pathway, which also plays an important role in autoimmune diseases [34] .
IL-17B
The role of IL-17B in fibrotic diseases is still limited in current research, and its specific function has not been fully elucidated. Existing studies have shown that in a mouse model of bleomycin induced pulmonary fibrosis, the absence of IL-17B can significantly slow down the progression of fibrosis [35] . In addition, another study suggests that the outer membrane vesicles produced by symbiotic microorganisms can induce the expression of IL-17B, activate neutrophil chemotaxis genes and Th17 cell activation genes, ultimately exacerbating lung inflammation and fibrosis [2] . However, some studies support the possibility that IL-17A and IL-17B have similar roles in regulating inflammation and fibrosis. For example, IL-17B can stimulate peritoneal neutrophils, macrophages, and lymphocytes to produce IL-6, IL-23, and IL-1 α [23] . In addition, it also regulates the release of TNF-α and IL-1 β from human monocytes/macrophages. IL-17B promotes the recruitment of cells expressing chemokine receptors CXCR4 or CXCR5, while experimental intraperitoneal administration of recombinant human IL-17B confirms its chemotactic appeal to neutrophils, who subsequently release chemokines for other cells. Not only that, IL-17B can also work synergistically with IL-33 to regulate the immune response mediated by helper T cells (Th) [36] . These findings suggest that IL-17B may promote fibrosis in the early stages of inflammation through its pro-inflammatory properties.
IL-17C
IL-17C is a multifunctional cytokine expressed in CD4+T cells, dendritic cells (DCs), macrophages, and epithelial cells, involved in the immune defense and inflammatory response of the lungs. These cells produce this interleukin during antibacterial activity, enhancing the inflammatory response [37] . In pulmonary fibrosis and certain lung diseases, IL-17C can act in conjunction with IL-17A or independently mediate fibrosis signaling.IL-17C works by binding to a heterodimeric receptor complex composed of IL-17RA and IL-17RE subunits. Among them, IL-17RE is mainly distributed on the surface of epithelial cells and Th17 cells. Th17 cells produce IL-17A and IL-17F upon stimulation by IL-17C, indicating that IL-17C may regulate the initial stage of the inflammatory cascade [38] . In addition, IL-17C triggers the NF-κ B/Act1 signaling axis upon binding to receptors, thereby activating the MAPK pathway to regulate target gene expression [39] . In the study of idiopathic pulmonary fibrosis (IPF), lipopolysaccharide induced lung injury was used as the IPF model [40] . In this study, IL-17C was confirmed to play a crucial role in pulmonary inflammation by mediating epithelial injury, pro-inflammatory cytokine release, and neutrophil recruitment. Recently, it has been reported that IL-17C exacerbates pulmonary fibrosis damage by driving neutrophilic inflammation in a model of influenza induced lung inflammation induced by Haemophilus influenzae and cigarette smoke [41] . These findings collectively reveal the pivotal role of IL-17C in inflammation fibrosis transition.
IL-17D
Little is currently known about the receptor and function of IL-17D. Research has shown that IL-17D can be detected in B lymphocytes and resting CD4+T cells. Although IL-17D has a weak activation effect on immune cells, it can regulate the secretion of pro-inflammatory cytokines in endothelial cells [42] . At present, there is still limited understanding of the role of IL-17D in the regulation of pulmonary fibrosis, and further research is needed to elucidate the relationship between IL-17D induced inflammatory response and the development of pulmonary fibrosis.
IL-17E
Recent studies have shown that the levels of IL-17E and its receptor IL-17RB are elevated in lung tissue of IPF patients, particularly in alveolar epithelial cells and lung fibroblasts, showing significant enrichment. IL-17E drives pulmonary fibrosis by mediating EMT of alveolar epithelial cells and recruiting and activating pulmonary fibroblasts [43] . It is interesting that this is related to the release of IL-13, which exacerbates collagen deposition during the IPF process. Specifically, IL-25 promotes IL-13 release by activating type 2 innate lymphocytes (ILC2s), thereby triggering collagen deposition during IPF [44] .
IL-17F
IL-17F and IL-17A have high sequence homology and are co expressed in the same cell population. However, there are significant differences in the roles of these two cytokines in fibrotic diseases [45] . IL-17A and IL-17F both have pro-inflammatory and pro fibrotic effects in pulmonary fibrosis, but IL-17A has a more significant effect. The effect of IL-17F may be relatively weak, but it may synergize with IL-17A in certain specific inflammatory environments. In addition, IL-17F transmits signals through the same IL-17RA/RC receptor complex, but with lower receptor affinity, resulting in weaker downstream signal intensity. Previous studies have shown that IL-17F activates neutrophils and regulates IL-17A related inflammatory gene expression through signaling components such as IL-17RA, Act1, and TRAF6, which have been shown to be key participants in IL-17A mediated inflammatory responses. Animal experiments have shown that specific overexpression of IL-17F in mouse lung tissue leads to infiltration of macrophages and lymphocytes, as well as increased mucus secretion. Although there is currently no direct evidence to suggest that IL-17F contributes to the progression of idiopathic pulmonary fibrosis , these observations related to IL-17A and inflammatory response suggest that IL-17F may be a potential effective target for treating IPF.
The molecular mechanism of IL-17 in pulmonary fibrosis
Pulmonary fibrosis is a complex pathological process with unknown etiology, which may be related to long-term exposure of the lungs to various types and sources of damaging factors. This process begins with damage to the alveolar epithelium, followed by a series of coagulation reactions, inflammatory reactions, and abnormal remodeling and regeneration of lung tissue, ultimately leading to progressive proliferation of fibrous connective tissue. Although studies have shown that environmental factors such as smoking and ionizing radiation may be key triggers [46] , the potential molecular mechanisms underlying the development of these diseases are still not fully understood. At the same time, inflammation and its role in the proliferation phase are evident, and the intercellular regulatory system gradually shifts towards promoting collagen synthesis, fibroblast proliferation, activation, and differentiation. This process involves the epithelial mesenchymal transition mechanism and excessive deposition of extracellular matrix components [47] .
The cytokines of the IL-17 family have pleiotropy and exert effective and diverse functions in vivo through classical and non classical signaling pathways [48] . Classical signal transduction induces transcription and post transcriptional mechanisms, which involve autoimmunity, hypersensitivity reactions, and metabolic reprogramming in lymphoid tissue. Non classical signal transduction collaborates with other receptor systems to regulate tissue repair and regeneration. According to classical concepts, pulmonary fibrosis is a gradual process that transitions from the initial injury stage to the inflammatory response stage, followed by the repair, regeneration, and remodeling of lung tissue. Genetic susceptibility factors, such as polymorphisms in Toll like receptor interacting protein genes, may participate in disease initiation by inhibiting the TGF-β signaling pathway [49] .
IL-17 binds to the IL-17 receptor and initiates signal transduction by recruiting the adaptor protein Act1 through the SEFIR domain. Act1 rapidly recruits and ubiquitinates TNF receptor associated factor 6 , activates various downstream pathways, and induces transcription of IL-17A target genes [50] . These pathways include: TRAF6/TAK1/NF-κB pathway, TRAF6/MAPK/AP-1 pathway, and Act1/TRAF6/C/EBP pathway (Figure 1) [51] .
Figure 1. IL-17 molecular mechanism pathway.Note: Act1 NF-κB activator 1; AP-1 activator protein-1; C/EBP CCAAT/enhancer-binding protein; MAPK: mitogen-activated protein kinase; NF-κB nuclear factor-kappaB; TRAF6 tumor necrosis factor receptor associated factor 6; TAK1 transforming growth factor-beta-activated kinase 1
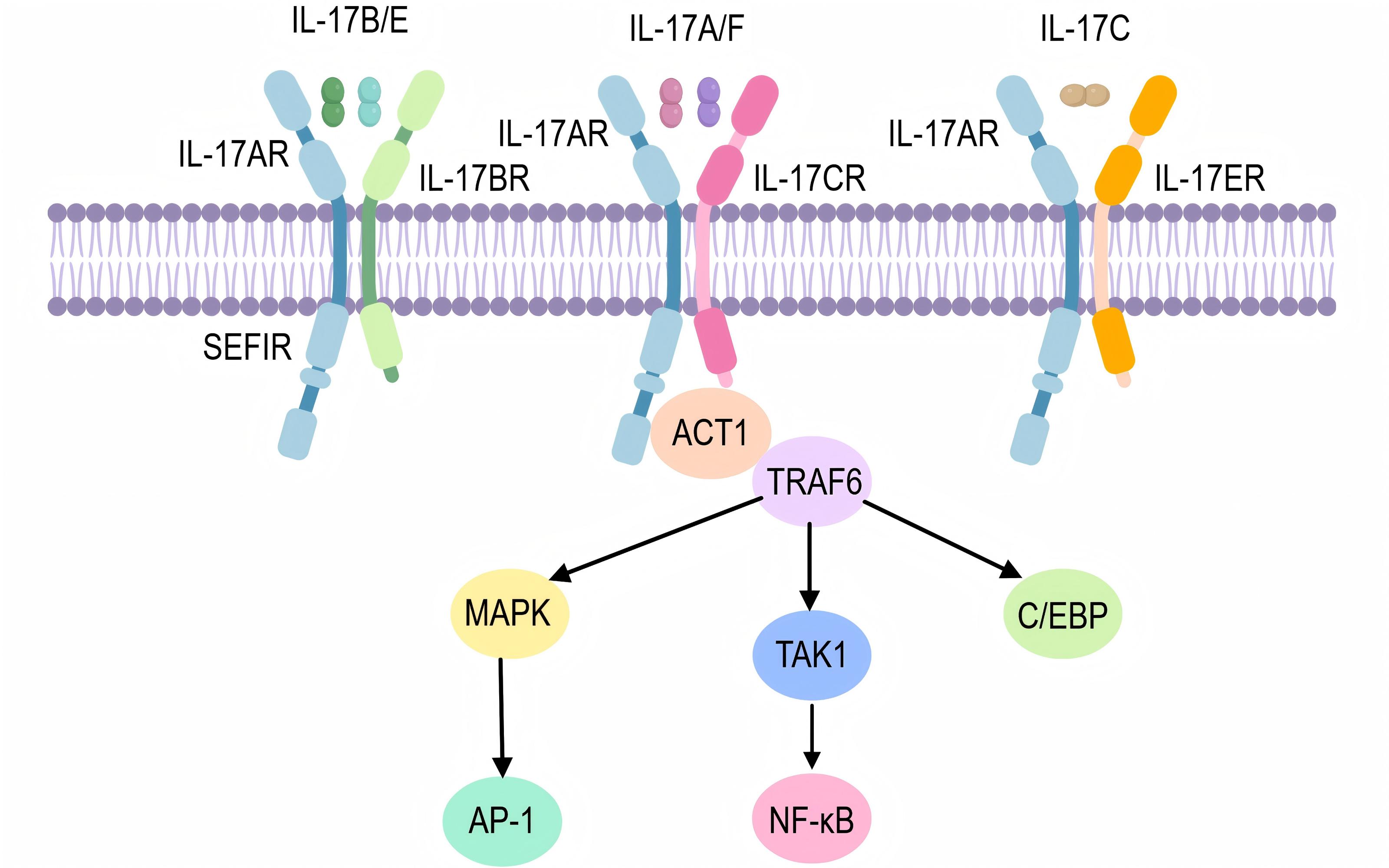
Act1 is a non redundant activator of IL-17RA dependent signaling, acting as a lysine-63 (K63) E3 ubiquitin ligase, recruiting and ubiquitinating TRAF6, activating the complex of transforming growth factor beta activated kinase 1 (TAK1) and nuclear factor kappa B inhibitor kinase (IKK) [52] . Then, IKK phosphorylates the NF-κ B subunit, and the labeled I κ B is hydrolyzed by proteases, exposing the nuclear localization signal of NF-κ B. This signal is released and binds to the target gene to promote its transcription. In the nucleus, NF-k B directly or in combination with activator protein-1 (AP-1) induces transcription of target genes [53] . After activation of these signaling pathways, they first promote the expression of antimicrobial peptides, chemokines, as well as cytokines such as TNF-α, IL-1 β, IL-6, and IL-23 in host cells. Subsequently, these inflammatory mediators recruit pro-inflammatory immune cells such as neutrophils and Th17 cells, ultimately forming a persistent inflammatory positive feedback loop [54] .
Targeted therapy
Given the strong pro-inflammatory effect of IL-17, drugs targeting IL-17 or IL-17R are potential therapeutic candidates for inflammatory autoimmune diseases [55] . Clinical trials have shown that antibodies targeting IL-12p40 (ustekinumab), IL-17A (sukinumab and izumab), IL-17A and IL-17F (bimekizumab), IL-17RA (brodalumab), and IL-23 (guselkumab, tildrakizumab, and risankizumab) are effective in treating moderate to severe psoriasis [56] . Yiqizhu monoclonal antibody is a humanized IgG4 monoclonal antibody that specifically binds to IL-17A to block its interaction with receptors and inhibit the release of pro-inflammatory cytokines. Research has found that dual antibodies targeting IL-17A/F (compared to monoclonal antibodies) show better efficacy in treating psoriasis than single target inhibitors, but these drugs have poor therapeutic response in rheumatoid arthritis (RA) and multiple sclerosis (MS) [57] .
Further research reveals that abnormal activation of the Act1-SHP2 complex leads to sustained IL-17 signaling and triggers treatment resistance. Animal experiments have shown that SHP2 inhibitors and small molecules that disrupt Act1-SHP2 interactions (such as Igulamide) can effectively alleviate inflammatory responses in mouse MS and RA models [58] . In the exploration of coronavirus disease 2019 (COVID-19 ) treatment, patients treated with netakimab monoclonal antibody showed reduced lung lesion size and decreased oxygen therapy demand, and improved survival rates [59] . However, in another study, netakimab treatment reduced C-reactive protein levels and improved some clinical parameters, but did not reduce the need for mechanical ventilation or improve the survival rate of COVID-19 patients [60] . These and other studies suggest that transient inhibition of IL-17 may be a therapeutic option for controlling excessive inflammation during acute viral infections.
In the field of respiratory diseases, IL-17 is involved in the pathogenesis of corticosteroid resistant neutropenic asthma. The experimental model of allergic asthma in mice has confirmed that anti-IL-17 monoclonal antibody can reverse neutrophil infiltration in the lungs [56] . Clinical observations have found that IL-17 levels and Th17 cells are increased in patients with neutropenic asthma [61] . Although clinical trials have not yet demonstrated a significant positive effect of blocking the IL-17 pathway on human asthma, patient stratification in future trials may improve outcomes [62] .
However, all monoclonal antibodies targeting the IL-17-IL-17R pathway and approved for treatment have some drawbacks, such as requiring injection administration, poor tissue permeability, and various adverse reactions that escalate immune system inflammatory responses. To this end, researchers are developing novel oral small molecule drugs (SMD) that regulate immune responses by blocking IL-17A/IL-17RA protein interactions [63] . These small molecule drugs have advantages in terms of production cost, delivery convenience, and potential higher efficacy.
Conclusion
IL-17 plays an important role in the occurrence and development of pulmonary fibrosis. It can promote inflammatory response and fibrosis process, becoming a potential therapeutic target. At present, there are multiple treatment methods targeting IL-17 under research, but their efficacy and safety still need further clinical trial verification. Future research needs to further explore the mechanism of IL-17 in pulmonary fibrosis, search for more effective treatment methods, and provide new ideas and strategies for the treatment of pulmonary fibrosis.
Abbreviations
Act1: NF-κB Activator 1; Act1-SHP2: Act1-Tyrosine Phosphatase; AP-1: Activator Protein-1;α-SMA: alpha-smooth muscle actin; BLM: Bleomycin ; CTLA-8: Cytotoxic T lymphocyte Associated Antigen 8; C/EBP: CCAAT/Enhancer-Binding Protein; CXCL1/2/5: Chemokine (C-X-C Motif) Ligand 1/2/5; CSF2: Colony Stimulating Factor 2; CCL1: Chemokine (C-X-C Motif) Ligand 1; CXCR4/5: Chemokine receptor 4/5; COVID-19: Coronavirus Disease 2019; DC: Dendritic Cells; EMT: Epithelial Mesenchymal Transition; FAD: Food and Drug Administration; IL-17: Interleukin 17; IL-17A/B/C/D/E/F: Interleukin 17A/B/C/D/E/F; IL-17RA/RB/RC/RD/RE/RF: IL-17 receptor A/B/C/D/E/F; ILD: Interstitial Lung Disease; IPF: Idiopathic Pulmonary Fibrosis; IL-17R: The IL-17 receptor; IL-1/6/8/23: Interleukin 1/6/8/23; iNKT: invariant Natural Killer T cells; ILC: Innate Lymphocytes; ILC2/3: Type 2/3 Innate Lymphoid Cells; IL-12p40: Interleukin 12 Subunit p40; IKK: Inhibitor Of KappaB Kinase ; MAPK: Mitogen-Activated Protein Kinase; MS: multiple sclerosis; NF-κB: Nuclear; Factor-KappaB; NK: Natural Killer; RA: Rheumatoid Arthritis; SMD: small molecule drugs; TRAF6: Tumor Necrosis Factor Receptor Associated Factor 6; TNF: Tumor Necrosis Factor; TNF-α: Tumor Necrosis Factor alpha; TGF-β: Transforming Growth Factor beta; TAK1: Transforming growth factor-beta-activated kinase 1
Declarations
Author Contributions
Ge Zhou(First Author): writing original draft, prepare, create, or express the content for publication, especially in writing the initial draft, including substantive translation.
Amin Li: writing review and editing, prepare, create, or express the content for publication, especially in writing the initial draft, including substantive translation.
Ruikai Wang(Corresponding Author): supervision, supervise and lead the planning and execution of research activities.
All authors read and approved the final manuscript.
Acknowledgements
Not Applicable.
Funding Information
Funding: This work was supported by Health Research Project of Anhui Province (grant No. AHWJ2024BAd30021), Scientific Research Foundation for High-Level Talents of Anhui University of Science and Technology (grant No. 2021yjrc14) and Graduate Innovation Fund Project of Anhui University of Science and Technology in 2025 (grant No.2025cx2140).
Ethics Approval and Consent to Participate
Not Applicable.
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
All data needed to evaluate the conclusions in the paper are present in the paper or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
References
[1]Bridges JP, Vladar EK, Kurche JS, Krivoi A, Stancil IT, Dobrinskikh E, et al. (2025). Progressive lung fibrosis: reprogramming a genetically vulnerable bronchoalveolar epithelium. Journal of Clinical Investigation, 135(1). https://doi.org/10.1172/JCI183836
[2]Chen T, Sun W, Xu Z-j. (2024). The immune mechanisms of acute exacerbations of idiopathic pulmonary fibrosis. Frontiers in Immunology, 15. https://doi.org/10.3389/fimmu.2024.1450688
[3]Mackintosh JA, Keir G, Troy LK, Holland AE, Grainge C, Chambers DC, et al. (2024). Treatment of idiopathic pulmonary fibrosis and progressive pulmonary fibrosis: A position statement from the Thoracic Society of Australia and New Zealand 2023 revision. Respirology, 29(2):105-35. https://doi.org/10.1111/resp.14656
[4]Han X, Su X, Che M, Liu L, Nie P, Wang S. (2025). Identification and Expression Analyses of IL-17/IL-17R Gene Family in Snakehead (Channa argus) Following Nocardia seriolae Infection. Genes, 16(3). https://doi.org/10.3390/genes16030253
[5]Huangfu L, Li R, Huang Y, Wang S. (2023). The IL-17 family in diseases: from bench to bedside. Signal Transduction and Targeted Therapy, 8(1). https://doi.org/10.1038/s41392-023-01620-3
[6]Chen S, Fan H, Ran C, Hong Y, Feng H, Yue Z, et al. (2024). The IL-17 pathway intertwines with neurotrophin and TLR/IL-1R pathways since its domain shuffling origin. Proceedings of the National Academy of Sciences of the United States of America, 121(19). https://doi.org/10.1073/pnas.2400903121
[7]Kolls JK, Lindén A. (2004). Interleukin-17 Family Members and Inflammation. Immunity, 21(4):467-76. https://doi.org/10.1016/j.immuni.2004.08.018
[8]Nie Y-J, Wu S-H, Xuan Y-H, Yan G. (2022). Role of IL-17 family cytokines in the progression of IPF from inflammation to fibrosis. Military Medical Research, 9(1). https://doi.org/10.1186/s40779-022-00382-3
[9]Tollenaere MAX, Hebsgaard J, Ewald DA, Lovato P, Garcet S, Li X, et al. (2021). Signalling of multiple interleukin (IL)‐17 family cytokines via IL‐17 receptor A drives psoriasis‐related inflammatory pathways. British Journal of Dermatology, 185(3):585-94. https://doi.org/10.1111/bjd.20090
[10]Wen Y, Chen Q, Wang H, Xie S, Chen H, Yao W, et al. (2024). Contribution of IL-17C-mediated macrophage polarization to Type 17 inflammation in neutrophilic asthma. Cell Communication and Signaling, 22(1). https://doi.org/10.1186/s12964-024-01937-8
[11]Mu X, Gu R, Tang M, Wu X, He W, Nie X. (2024). IL-17 in wound repair: bridging acute and chronic responses. Cell Communication and Signaling, 22(1). https://doi.org/10.1186/s12964-024-01668-w
[12]Knizkova D, Pribikova M, Draberova H, Semberova T, Trivic T, Synackova A, et al. (2022). CMTM4 is a subunit of the IL-17 receptor and mediates autoimmune pathology. Nature Immunology, 23(11):1644-52. https://doi.org/10.1038/s41590-022-01325-9
[13]Herjan T, Hong L, Bubenik J, Bulek K, Qian W, Liu C, et al. (2018). IL-17-receptor-associated adaptor Act1 directly stabilizes mRNAs to mediate IL-17 inflammatory signaling. Nature Immunology, 19(4):354-65. https://doi.org/10.1038/s41590-018-0071-9
[14]Brackman LC, Jung MS, Green EH, Joshi N, Revetta FL, McClain MS, et al. (2024)IL-17 signaling protects against Helicobacter pylori- induced gastric cancer. Gut Microbes, 16(1).https://doi.org/10.1080/19490976.2024.2430421
[15]Enzel D, Kriventsov M, Sataieva T, Malygina V. (2024). Cellular and Molecular Genetic Mechanisms of Lung Fibrosis Development and the Role of Vitamin D: A Review. International Journal of Molecular Sciences, 25(16). https://doi.org/10.3390/ijms25168946
[16]Meehan EV, Wang K. (2022). Interleukin-17 Family Cytokines in Metabolic Disorders and Cancer. Genes, 13(9). https://doi.org/10.3390/genes13091643
[17]Gouda MM, Bhandary YP. (2019). Acute Lung Injury: IL-17A-Mediated Inflammatory Pathway and Its Regulation by Curcumin. Inflammation, 42(4), 1160–1169. https://doi.org/10.1007/s10753-019-01010-4
[18]Wilson MS, Madala SK, Ramalingam TR, Gochuico BR, Rosas IO, Cheever AW, et al. (2010). Bleomycin and IL-1beta-mediated pulmonary fibrosis is IL-17A dependent. Journal of experimental medicine, 207(3), 535–552. https://doi.org/10.1084/jem.20092121
[19]Liu C, Zhu L, Fukuda K, Ouyang S, Chen X, Wang C, et al. (2017). The flavonoid cyanidin blocks binding of the cytokine interleukin-17A to the IL-17RA subunit to alleviate inflammation in vivo. Science Signaling, 10(467). https://doi.org/10.1126/scisignal.aaf8823
[20]Golebski K, Ros XR, Nagasawa M, van Tol S, Heesters BA, Aglmous H, et al. (2019). IL-1β, IL-23, and TGF-β drive plasticity of human ILC2s towards IL-17-producing ILCs in nasal inflammation. Nature Communications, 10(1). https://doi.org/10.1038/s41467-019-09883-7
[21]Glatt S, Baeten D, Baker T, Griffiths M, Ionescu L, Lawson ADG, et al. (2018). Dual IL-17A and IL-17F neutralisation by bimekizumab in psoriatic arthritis: evidence from preclinical experiments and a randomised placebo-controlled clinical trial that IL-17F contributes to human chronic tissue inflammation. Annals of the Rheumatic Diseases, 77(4):523-32. https://doi.org/10.1136/annrheumdis-2017-212127
[22]Chang Seon H, Reynolds Joseph M, Pappu Bhanu P, Chen G, Martinez Gustavo J, Dong C. (2011). Interleukin-17C Promotes Th17 Cell Responses and Autoimmune Disease via Interleukin-17 Receptor E. Immunity, 35(4):611-21. https://doi.org/10.1016/j.immuni.2011.09.010
[23]Reynolds Joseph M, Lee Y-H, Shi Y, Wang X, Angkasekwinai P, Nallaparaju Kalyan C, et al. (2015). Interleukin-17B Antagonizes Interleukin-25-Mediated Mucosal Inflammation. Immunity, 42(4):692-703. https://doi.org/10.1016/j.immuni.2015.03.008
[24]Shabgah AG, Fattahi E, Shahneh FZ. (2014). Interleukin-17 in human inflammatory diseases. Advances in Dermatology and Allergology, 31 (4):256-61. https://doi.org/10.5114/pdia.2014.40954
[25]Khader S, Gasse P, Riteau N, Vacher R, Michel M-L, Fautrel A, et al. (2011). IL-1 and IL-23 Mediate Early IL-17A Production in Pulmonary Inflammation Leading to Late Fibrosis. PLoS ONE, 6(8). https://doi.org/10.1371/journal.pone.0023185
[26]He Q, Cao J, Zhang M, Feng C. (2024). IL-17 in plasma and bronchoalveolar lavage fluid in non-neutropenic patients with invasive pulmonary aspergillosis. Frontiers in Cellular and Infection Microbiology, 14. https://doi.org/10.3389/fcimb.2024.1402888
[27]Jiang G, Liu CT, Zhang WD. (2018). IL‑17A and GDF15 are able to induce epithelial‑mesenchymal transition of lung epithelial cells in response to cigarette smoke. Experimental and Therapeutic Medicine, 16(1), 12-20. https://doi.org/10.3892/etm.2018.6145
[28]Sisto M, Lorusso L, Tamma R, Ingravallo G, Ribatti D, Lisi S. (2019). Interleukin-17 and -22 synergy linking inflammation and EMT-dependent fibrosis in Sjögren’s syndrome. Clinical and Experimental Immunology. 2019;198(2):261-72. https://doi.org/10.1111/cei.13337
[29]Qu Z, Dou W, Zhang K, Duan L, Zhou D, Yin S. (2022). IL-22 inhibits bleomycin-induced pulmonary fibrosis in association with inhibition of IL-17A in mice. Arthritis Research & Therapy, 24(1). https://doi.org/10.1186/s13075-022-02977-6
[30]Sisto M, Lisi S. (2023). Targeting Interleukin-17 as a Novel Treatment Option for Fibrotic Diseases. Journal of Clinical Medicine, 13(1). https://doi.org/10.3390/jcm13010164
[31]Liang M, Wang J, Chu H, Zhu X, He H, Liu Q, et al. (2013). Interleukin-22 Inhibits Bleomycin-Induced Pulmonary Fibrosis. Mediators of Inflammation,2013:1-11. https://doi.org/10.1155/2013/209179
[32]Gurczynski SJ, Moore BB. (2018). IL-17 in the lung: the good, the bad, and the ugly. American journal of physiology. Lung cellular and molecular physiology, 314(1), L6–L16. https://doi.org/10.1152/ajplung.00344.2017
[33]Dong Z, Yang Y, Zhang T, Li Y, Kang Q, Lei W,et al. (2013). siRNA-Act1 inhibits the function of IL-17 on lung fibroblasts via the NF-κB pathway. Respiration; international review of thoracic diseases, 86(4), 332–340. https://doi.org/10.1159/000348403
[34]Sønder SU, Saret S, Tang W, Sturdevant DE, Porcella SF, Siebenlist U. (2011). IL-17-induced NF-κB Activation via CIKS/Act1. Journal of Biological Chemistry, 286(15):12881-90. https://doi.org/10.1074/jbc.M110.199547
[35]Huang L. (2024). The role of IL-17 family cytokines in cardiac fibrosis. Frontiers in cardiovascular medicine, 11. https://doi.org/10.3389/fcvm.2024.1470362
[36]Morrow KN, Coopersmith CM, Ford ML. (2019). IL-17, IL-27, and IL-33: A Novel Axis Linked to Immunological Dysfunction During Sepsis. Frontiers in Immunology, 10. https://doi.org/10.3389/fimmu.2019.01982
[37]Krohn S, Nies JF, Kapffer S, Schmidt T, Riedel JH, Kaffke A, et al. (2018). IL-17C/IL-17 Receptor E Signaling in CD4+ T Cells Promotes TH17 Cell-Driven Glomerular Inflammation. Journal of the American Society of Nephrology : JASN, 29(4), 1210–1222. https://doi.org/10.1681/ASN.2017090949
[38]Vandeghinste N, Klattig J, Jagerschmidt C, Lavazais S, Marsais F, Haas JD, et al. (2018). Neutralization of IL-17C Reduces Skin Inflammation in Mouse Models of Psoriasis and Atopic Dermatitis. Journal of Investigative Dermatology, 138(7):1555-63. https://doi.org/10.1016/j.jid.2018.01.036
[39]Naglik JR, Conti HR, Whibley N, Coleman BM, Garg AV, Jaycox JR, et al. (2015). Signaling through IL-17C/IL-17RE Is Dispensable for Immunity to Systemic, Oral and Cutaneous Candidiasis. Plos One, 10(4). https://doi.org/10.1371/journal.pone.0122807
[40]Chen S, Zhang X, Yang C, Wang S, Shen H. (2022). Essential role of IL-17 in acute exacerbation of pulmonary fibrosis induced by non-typeable Haemophilus influenzae. Theranostics, 12(11):5125-37. https://doi.org/10.7150/thno.74809
[41]Yildirim AÖ, Vella G, Ritzmann F, Wolf L, Kamyschnikov A, Stodden H, et al. (2021). IL-17C contributes to NTHi-induced inflammation and lung damage in experimental COPD and is present in sputum during acute exacerbations. Plos One, 16(1). https://doi.org/10.1371/journal.pone.0243484
[42]Liu X, Sun S, Liu D. (2020). IL-17D: A Less Studied Cytokine of IL-17 Family. International Archives of Allergy and Immunology, 181(8):618-23. https://doi.org/10.1159/000508255
[43]Xu X, Luo S, Li B, Dai H, Zhang J. (2019). IL-25 contributes to lung fibrosis by directly acting on alveolar epithelial cells and fibroblasts. Experimental Biology and Medicine, 244(9):770-80. https://doi.org/10.1177/1535370219843827
[44]Hams E, Armstrong ME, Barlow JL, Saunders SP, Schwartz C, Cooke G, et al. (2013). IL-25 and type 2 innate lymphoid cells induce pulmonary fibrosis. Proceedings of the National Academy of Sciences, 111(1):367-72. https://doi.org/10.1073/pnas.1315854111
[45]Ritzmann F, Lunding LP, Bals R, Wegmann M, Beisswenger C. (2022). IL-17 Cytokines and Chronic Lung Diseases. Cells, 11(14). https://doi.org/10.3390/cells11142132
[46]Bellou V, Belbasis L, Evangelou E. (2021). Tobacco Smoking and Risk for Pulmonary Fibrosis. Ches, 160(3):983-93. https://doi.org/10.1016/j.chest.2021.04.035
[47]Sgalla G, Iovene B, Calvello M, Ori M, Varone F, Richeldi L. (2018). Idiopathic pulmonary fibrosis: pathogenesis and management. Respiratory Research,19(1). https://doi.org/10.1186/s12931-018-0730-2
[48]Li X, Bechara R, Zhao J, McGeachy MJ, Gaffen SL. (2019). IL-17 receptor–based signaling and implications for disease. Nature Immunology, 20(12):1594-602. https://doi.org/10.1038/s41590-019-0514-y
[49]McElroy AN, Invernizzi R, Laskowska JW, O'Neill A, Doroudian M, Moghoofei M, et al. (2022). Candidate Role for Toll-like Receptor 3 L412F Polymorphism and Infection in Acute Exacerbation of Idiopathic Pulmonary Fibrosis. American journal of respiratory and critical care medicine, 205(5), 550–562. https://doi.org/10.1164/rccm.202010-3880OC
[50]Theune WC, Chen J, Theune EV, Ye X, Ménoret A, Vella AT, et al. (2024). Interleukin-17 directly stimulates tumor infiltrating Tregs to prevent cancer development. Frontiers in Immunology, 15. https://doi.org/10.3389/fimmu.2024.1408710
[51]Li G, Chen H, Liu L, Xiao P, Xie Y, Geng X, et al. (2021). Role of Interleukin-17 in Acute Pancreatitis. Frontiers in Immunology,12. https://doi.org/10.3389/fimmu.2021.674803
[52]Zwicky P, Unger S, Becher B. (2020). Targeting interleukin-17 in chronic inflammatory disease: A clinical perspective. Journal of Experimental Medicine, 217(1).
[53]Ladjevac N, Milovanovic M, Jevtovic A, Arsenijevic D, Stojanovic B, Dimitrijevic Stojanovic M, et al. (2023). The Role of IL-17 in the Pathogenesis of Oral Squamous Cell Carcinoma. International Journal of Molecular Sciences, 24(12). https://doi.org/10.3390/ijms24129874
[54]Hadian Y, Bagood MD, Dahle SE, Sood A, Isseroff RR. (2019). Interleukin-17: Potential Target for Chronic Wounds. Mediators of inflammation, 2019. https://doi.org/10.1155/2019/1297675
[55]Flemming A. (2023). Why do IL-17-targeted therapies have limited efficacy?. Nature Reviews Immunology, 23(9), 543-543. https://doi.org/10.1038/s41577-023-00930-5
[56]Mills KHG. (2022). IL-17 and IL-17-producing cells in protection versus pathology. Nature reviews. Immunology, 23(1), 38–54. https://doi.org/10.1038/s41577-022-00746-9
[57]Wang J, Wang C, Liu L, Hong S, Ru Y, Sun X, et al. (2023). Adverse events associated with anti-IL-17 agents for psoriasis and psoriatic arthritis: a systematic scoping review. Frontiers in Immunology,14. https://doi.org/10.3389/fimmu.2023.993057
[58]Luo Q, Liu Y, Shi K, Shen X, Yang Y, Liang X, et al. (2023). An autonomous activation of interleukin-17 receptor signaling sustains inflammation and promotes disease progression. Immunity, 56(9):2006-20.e6. https://doi.org/10.1016/j.immuni.2023.06.012
[59]Maslennikov R, Ivashkin V, Vasilieva E, Chipurik M, Semikova P, Semenets V, et al. (2021).Interleukin 17 antagonist netakimab is effective and safe in the new coronavirus infection (COVID-19). European Cytokine Network, 32(1):8-14. https://doi.org/10.1684/ecn.2021.0463
[60]Avdeev SN, Trushenko NV, Tsareva NA, Yaroshetskiy AI, Merzhoeva ZM, Nuralieva GS, et al. (2021). Anti-IL-17 monoclonal antibodies in hospitalized patients with severe COVID-19: A pilot study. Cytokine, 146. https://doi.org/10.1016/j.cyto.2021.155627
[61]Wei Q, Liao J, Jiang M, Liu J, Liang X, Nong G. (2021). Relationship between Th17-mediated immunity and airway inflammation in childhood neutrophilic asthma. Allergy, Asthma & Clinical Immunology, 17(1). https://doi.org/10.1186/s13223-020-00504-3
[62]Xie Y, Abel PW, Casale TB, Tu Y. (2022). TH17 cells and corticosteroid insensitivity in severe asthma. Journal of Allergy and Clinical Immunology, 149(2):467-79. https://doi.org/10.1016/j.jaci.2021.12.769
[63]Zhang B, Dömling A. (2022). Small molecule modulators of IL-17A/IL-17RA: a patent review (2013-2021). Expert Opinion on Therapeutic Patents, 32(11):1161-73. https://doi.org/10.1080/13543776.2022.2143264
 Figures
Figures References
References Peer
Peer Information
InformationFigure 1. IL-17 molecular mechanism pathway.Note: Act1 NF-κB activator 1; AP-1 activator protein-1; C/EBP CCAAT/enhancer-binding protein; MAPK: mitogen-activated protein kinase; NF-κB nuclear factor-kappaB; TRAF6 tumor necrosis factor receptor associated factor 6; TAK1 transforming growth factor-beta-activated kinase 1

[1]Bridges JP, Vladar EK, Kurche JS, Krivoi A, Stancil IT, Dobrinskikh E, et al. (2025). Progressive lung fibrosis: reprogramming a genetically vulnerable bronchoalveolar epithelium. Journal of Clinical Investigation, 135(1). https://doi.org/10.1172/JCI183836
[2]Chen T, Sun W, Xu Z-j. (2024). The immune mechanisms of acute exacerbations of idiopathic pulmonary fibrosis. Frontiers in Immunology, 15. https://doi.org/10.3389/fimmu.2024.1450688
[3]Mackintosh JA, Keir G, Troy LK, Holland AE, Grainge C, Chambers DC, et al. (2024). Treatment of idiopathic pulmonary fibrosis and progressive pulmonary fibrosis: A position statement from the Thoracic Society of Australia and New Zealand 2023 revision. Respirology, 29(2):105-35. https://doi.org/10.1111/resp.14656
[4]Han X, Su X, Che M, Liu L, Nie P, Wang S. (2025). Identification and Expression Analyses of IL-17/IL-17R Gene Family in Snakehead (Channa argus) Following Nocardia seriolae Infection. Genes, 16(3). https://doi.org/10.3390/genes16030253
[5]Huangfu L, Li R, Huang Y, Wang S. (2023). The IL-17 family in diseases: from bench to bedside. Signal Transduction and Targeted Therapy, 8(1). https://doi.org/10.1038/s41392-023-01620-3
[6]Chen S, Fan H, Ran C, Hong Y, Feng H, Yue Z, et al. (2024). The IL-17 pathway intertwines with neurotrophin and TLR/IL-1R pathways since its domain shuffling origin. Proceedings of the National Academy of Sciences of the United States of America, 121(19). https://doi.org/10.1073/pnas.2400903121
[7]Kolls JK, Lindén A. (2004). Interleukin-17 Family Members and Inflammation. Immunity, 21(4):467-76. https://doi.org/10.1016/j.immuni.2004.08.018
[8]Nie Y-J, Wu S-H, Xuan Y-H, Yan G. (2022). Role of IL-17 family cytokines in the progression of IPF from inflammation to fibrosis. Military Medical Research, 9(1). https://doi.org/10.1186/s40779-022-00382-3
[9]Tollenaere MAX, Hebsgaard J, Ewald DA, Lovato P, Garcet S, Li X, et al. (2021). Signalling of multiple interleukin (IL)‐17 family cytokines via IL‐17 receptor A drives psoriasis‐related inflammatory pathways. British Journal of Dermatology, 185(3):585-94. https://doi.org/10.1111/bjd.20090
[10]Wen Y, Chen Q, Wang H, Xie S, Chen H, Yao W, et al. (2024). Contribution of IL-17C-mediated macrophage polarization to Type 17 inflammation in neutrophilic asthma. Cell Communication and Signaling, 22(1). https://doi.org/10.1186/s12964-024-01937-8
[11]Mu X, Gu R, Tang M, Wu X, He W, Nie X. (2024). IL-17 in wound repair: bridging acute and chronic responses. Cell Communication and Signaling, 22(1). https://doi.org/10.1186/s12964-024-01668-w
[12]Knizkova D, Pribikova M, Draberova H, Semberova T, Trivic T, Synackova A, et al. (2022). CMTM4 is a subunit of the IL-17 receptor and mediates autoimmune pathology. Nature Immunology, 23(11):1644-52. https://doi.org/10.1038/s41590-022-01325-9
[13]Herjan T, Hong L, Bubenik J, Bulek K, Qian W, Liu C, et al. (2018). IL-17-receptor-associated adaptor Act1 directly stabilizes mRNAs to mediate IL-17 inflammatory signaling. Nature Immunology, 19(4):354-65. https://doi.org/10.1038/s41590-018-0071-9
[14]Brackman LC, Jung MS, Green EH, Joshi N, Revetta FL, McClain MS, et al. (2024)IL-17 signaling protects against Helicobacter pylori- induced gastric cancer. Gut Microbes, 16(1).https://doi.org/10.1080/19490976.2024.2430421
[15]Enzel D, Kriventsov M, Sataieva T, Malygina V. (2024). Cellular and Molecular Genetic Mechanisms of Lung Fibrosis Development and the Role of Vitamin D: A Review. International Journal of Molecular Sciences, 25(16). https://doi.org/10.3390/ijms25168946
[16]Meehan EV, Wang K. (2022). Interleukin-17 Family Cytokines in Metabolic Disorders and Cancer. Genes, 13(9). https://doi.org/10.3390/genes13091643
[17]Gouda MM, Bhandary YP. (2019). Acute Lung Injury: IL-17A-Mediated Inflammatory Pathway and Its Regulation by Curcumin. Inflammation, 42(4), 1160–1169. https://doi.org/10.1007/s10753-019-01010-4
[18]Wilson MS, Madala SK, Ramalingam TR, Gochuico BR, Rosas IO, Cheever AW, et al. (2010). Bleomycin and IL-1beta-mediated pulmonary fibrosis is IL-17A dependent. Journal of experimental medicine, 207(3), 535–552. https://doi.org/10.1084/jem.20092121
[19]Liu C, Zhu L, Fukuda K, Ouyang S, Chen X, Wang C, et al. (2017). The flavonoid cyanidin blocks binding of the cytokine interleukin-17A to the IL-17RA subunit to alleviate inflammation in vivo. Science Signaling, 10(467). https://doi.org/10.1126/scisignal.aaf8823
[20]Golebski K, Ros XR, Nagasawa M, van Tol S, Heesters BA, Aglmous H, et al. (2019). IL-1β, IL-23, and TGF-β drive plasticity of human ILC2s towards IL-17-producing ILCs in nasal inflammation. Nature Communications, 10(1). https://doi.org/10.1038/s41467-019-09883-7
[21]Glatt S, Baeten D, Baker T, Griffiths M, Ionescu L, Lawson ADG, et al. (2018). Dual IL-17A and IL-17F neutralisation by bimekizumab in psoriatic arthritis: evidence from preclinical experiments and a randomised placebo-controlled clinical trial that IL-17F contributes to human chronic tissue inflammation. Annals of the Rheumatic Diseases, 77(4):523-32. https://doi.org/10.1136/annrheumdis-2017-212127
[22]Chang Seon H, Reynolds Joseph M, Pappu Bhanu P, Chen G, Martinez Gustavo J, Dong C. (2011). Interleukin-17C Promotes Th17 Cell Responses and Autoimmune Disease via Interleukin-17 Receptor E. Immunity, 35(4):611-21. https://doi.org/10.1016/j.immuni.2011.09.010
[23]Reynolds Joseph M, Lee Y-H, Shi Y, Wang X, Angkasekwinai P, Nallaparaju Kalyan C, et al. (2015). Interleukin-17B Antagonizes Interleukin-25-Mediated Mucosal Inflammation. Immunity, 42(4):692-703. https://doi.org/10.1016/j.immuni.2015.03.008
[24]Shabgah AG, Fattahi E, Shahneh FZ. (2014). Interleukin-17 in human inflammatory diseases. Advances in Dermatology and Allergology, 31 (4):256-61. https://doi.org/10.5114/pdia.2014.40954
[25]Khader S, Gasse P, Riteau N, Vacher R, Michel M-L, Fautrel A, et al. (2011). IL-1 and IL-23 Mediate Early IL-17A Production in Pulmonary Inflammation Leading to Late Fibrosis. PLoS ONE, 6(8). https://doi.org/10.1371/journal.pone.0023185
[26]He Q, Cao J, Zhang M, Feng C. (2024). IL-17 in plasma and bronchoalveolar lavage fluid in non-neutropenic patients with invasive pulmonary aspergillosis. Frontiers in Cellular and Infection Microbiology, 14. https://doi.org/10.3389/fcimb.2024.1402888
[27]Jiang G, Liu CT, Zhang WD. (2018). IL‑17A and GDF15 are able to induce epithelial‑mesenchymal transition of lung epithelial cells in response to cigarette smoke. Experimental and Therapeutic Medicine, 16(1), 12-20. https://doi.org/10.3892/etm.2018.6145
[28]Sisto M, Lorusso L, Tamma R, Ingravallo G, Ribatti D, Lisi S. (2019). Interleukin-17 and -22 synergy linking inflammation and EMT-dependent fibrosis in Sjögren’s syndrome. Clinical and Experimental Immunology. 2019;198(2):261-72. https://doi.org/10.1111/cei.13337
[29]Qu Z, Dou W, Zhang K, Duan L, Zhou D, Yin S. (2022). IL-22 inhibits bleomycin-induced pulmonary fibrosis in association with inhibition of IL-17A in mice. Arthritis Research & Therapy, 24(1). https://doi.org/10.1186/s13075-022-02977-6
[30]Sisto M, Lisi S. (2023). Targeting Interleukin-17 as a Novel Treatment Option for Fibrotic Diseases. Journal of Clinical Medicine, 13(1). https://doi.org/10.3390/jcm13010164
[31]Liang M, Wang J, Chu H, Zhu X, He H, Liu Q, et al. (2013). Interleukin-22 Inhibits Bleomycin-Induced Pulmonary Fibrosis. Mediators of Inflammation,2013:1-11. https://doi.org/10.1155/2013/209179
[32]Gurczynski SJ, Moore BB. (2018). IL-17 in the lung: the good, the bad, and the ugly. American journal of physiology. Lung cellular and molecular physiology, 314(1), L6–L16. https://doi.org/10.1152/ajplung.00344.2017
[33]Dong Z, Yang Y, Zhang T, Li Y, Kang Q, Lei W,et al. (2013). siRNA-Act1 inhibits the function of IL-17 on lung fibroblasts via the NF-κB pathway. Respiration; international review of thoracic diseases, 86(4), 332–340. https://doi.org/10.1159/000348403
[34]Sønder SU, Saret S, Tang W, Sturdevant DE, Porcella SF, Siebenlist U. (2011). IL-17-induced NF-κB Activation via CIKS/Act1. Journal of Biological Chemistry, 286(15):12881-90. https://doi.org/10.1074/jbc.M110.199547
[35]Huang L. (2024). The role of IL-17 family cytokines in cardiac fibrosis. Frontiers in cardiovascular medicine, 11. https://doi.org/10.3389/fcvm.2024.1470362
[36]Morrow KN, Coopersmith CM, Ford ML. (2019). IL-17, IL-27, and IL-33: A Novel Axis Linked to Immunological Dysfunction During Sepsis. Frontiers in Immunology, 10. https://doi.org/10.3389/fimmu.2019.01982
[37]Krohn S, Nies JF, Kapffer S, Schmidt T, Riedel JH, Kaffke A, et al. (2018). IL-17C/IL-17 Receptor E Signaling in CD4+ T Cells Promotes TH17 Cell-Driven Glomerular Inflammation. Journal of the American Society of Nephrology : JASN, 29(4), 1210–1222. https://doi.org/10.1681/ASN.2017090949
[38]Vandeghinste N, Klattig J, Jagerschmidt C, Lavazais S, Marsais F, Haas JD, et al. (2018). Neutralization of IL-17C Reduces Skin Inflammation in Mouse Models of Psoriasis and Atopic Dermatitis. Journal of Investigative Dermatology, 138(7):1555-63. https://doi.org/10.1016/j.jid.2018.01.036
[39]Naglik JR, Conti HR, Whibley N, Coleman BM, Garg AV, Jaycox JR, et al. (2015). Signaling through IL-17C/IL-17RE Is Dispensable for Immunity to Systemic, Oral and Cutaneous Candidiasis. Plos One, 10(4). https://doi.org/10.1371/journal.pone.0122807
[40]Chen S, Zhang X, Yang C, Wang S, Shen H. (2022). Essential role of IL-17 in acute exacerbation of pulmonary fibrosis induced by non-typeable Haemophilus influenzae. Theranostics, 12(11):5125-37. https://doi.org/10.7150/thno.74809
[41]Yildirim AÖ, Vella G, Ritzmann F, Wolf L, Kamyschnikov A, Stodden H, et al. (2021). IL-17C contributes to NTHi-induced inflammation and lung damage in experimental COPD and is present in sputum during acute exacerbations. Plos One, 16(1). https://doi.org/10.1371/journal.pone.0243484
[42]Liu X, Sun S, Liu D. (2020). IL-17D: A Less Studied Cytokine of IL-17 Family. International Archives of Allergy and Immunology, 181(8):618-23. https://doi.org/10.1159/000508255
[43]Xu X, Luo S, Li B, Dai H, Zhang J. (2019). IL-25 contributes to lung fibrosis by directly acting on alveolar epithelial cells and fibroblasts. Experimental Biology and Medicine, 244(9):770-80. https://doi.org/10.1177/1535370219843827
[44]Hams E, Armstrong ME, Barlow JL, Saunders SP, Schwartz C, Cooke G, et al. (2013). IL-25 and type 2 innate lymphoid cells induce pulmonary fibrosis. Proceedings of the National Academy of Sciences, 111(1):367-72. https://doi.org/10.1073/pnas.1315854111
[45]Ritzmann F, Lunding LP, Bals R, Wegmann M, Beisswenger C. (2022). IL-17 Cytokines and Chronic Lung Diseases. Cells, 11(14). https://doi.org/10.3390/cells11142132
[46]Bellou V, Belbasis L, Evangelou E. (2021). Tobacco Smoking and Risk for Pulmonary Fibrosis. Ches, 160(3):983-93. https://doi.org/10.1016/j.chest.2021.04.035
[47]Sgalla G, Iovene B, Calvello M, Ori M, Varone F, Richeldi L. (2018). Idiopathic pulmonary fibrosis: pathogenesis and management. Respiratory Research,19(1). https://doi.org/10.1186/s12931-018-0730-2
[48]Li X, Bechara R, Zhao J, McGeachy MJ, Gaffen SL. (2019). IL-17 receptor–based signaling and implications for disease. Nature Immunology, 20(12):1594-602. https://doi.org/10.1038/s41590-019-0514-y
[49]McElroy AN, Invernizzi R, Laskowska JW, O'Neill A, Doroudian M, Moghoofei M, et al. (2022). Candidate Role for Toll-like Receptor 3 L412F Polymorphism and Infection in Acute Exacerbation of Idiopathic Pulmonary Fibrosis. American journal of respiratory and critical care medicine, 205(5), 550–562. https://doi.org/10.1164/rccm.202010-3880OC
[50]Theune WC, Chen J, Theune EV, Ye X, Ménoret A, Vella AT, et al. (2024). Interleukin-17 directly stimulates tumor infiltrating Tregs to prevent cancer development. Frontiers in Immunology, 15. https://doi.org/10.3389/fimmu.2024.1408710
[51]Li G, Chen H, Liu L, Xiao P, Xie Y, Geng X, et al. (2021). Role of Interleukin-17 in Acute Pancreatitis. Frontiers in Immunology,12. https://doi.org/10.3389/fimmu.2021.674803
[52]Zwicky P, Unger S, Becher B. (2020). Targeting interleukin-17 in chronic inflammatory disease: A clinical perspective. Journal of Experimental Medicine, 217(1).
[53]Ladjevac N, Milovanovic M, Jevtovic A, Arsenijevic D, Stojanovic B, Dimitrijevic Stojanovic M, et al. (2023). The Role of IL-17 in the Pathogenesis of Oral Squamous Cell Carcinoma. International Journal of Molecular Sciences, 24(12). https://doi.org/10.3390/ijms24129874
[54]Hadian Y, Bagood MD, Dahle SE, Sood A, Isseroff RR. (2019). Interleukin-17: Potential Target for Chronic Wounds. Mediators of inflammation, 2019. https://doi.org/10.1155/2019/1297675
[55]Flemming A. (2023). Why do IL-17-targeted therapies have limited efficacy?. Nature Reviews Immunology, 23(9), 543-543. https://doi.org/10.1038/s41577-023-00930-5
[56]Mills KHG. (2022). IL-17 and IL-17-producing cells in protection versus pathology. Nature reviews. Immunology, 23(1), 38–54. https://doi.org/10.1038/s41577-022-00746-9
[57]Wang J, Wang C, Liu L, Hong S, Ru Y, Sun X, et al. (2023). Adverse events associated with anti-IL-17 agents for psoriasis and psoriatic arthritis: a systematic scoping review. Frontiers in Immunology,14. https://doi.org/10.3389/fimmu.2023.993057
[58]Luo Q, Liu Y, Shi K, Shen X, Yang Y, Liang X, et al. (2023). An autonomous activation of interleukin-17 receptor signaling sustains inflammation and promotes disease progression. Immunity, 56(9):2006-20.e6. https://doi.org/10.1016/j.immuni.2023.06.012
[59]Maslennikov R, Ivashkin V, Vasilieva E, Chipurik M, Semikova P, Semenets V, et al. (2021).Interleukin 17 antagonist netakimab is effective and safe in the new coronavirus infection (COVID-19). European Cytokine Network, 32(1):8-14. https://doi.org/10.1684/ecn.2021.0463
[60]Avdeev SN, Trushenko NV, Tsareva NA, Yaroshetskiy AI, Merzhoeva ZM, Nuralieva GS, et al. (2021). Anti-IL-17 monoclonal antibodies in hospitalized patients with severe COVID-19: A pilot study. Cytokine, 146. https://doi.org/10.1016/j.cyto.2021.155627
[61]Wei Q, Liao J, Jiang M, Liu J, Liang X, Nong G. (2021). Relationship between Th17-mediated immunity and airway inflammation in childhood neutrophilic asthma. Allergy, Asthma & Clinical Immunology, 17(1). https://doi.org/10.1186/s13223-020-00504-3
[62]Xie Y, Abel PW, Casale TB, Tu Y. (2022). TH17 cells and corticosteroid insensitivity in severe asthma. Journal of Allergy and Clinical Immunology, 149(2):467-79. https://doi.org/10.1016/j.jaci.2021.12.769
[63]Zhang B, Dömling A. (2022). Small molecule modulators of IL-17A/IL-17RA: a patent review (2013-2021). Expert Opinion on Therapeutic Patents, 32(11):1161-73. https://doi.org/10.1080/13543776.2022.2143264
Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-02-14
Accepted 2025-05-26
Published 2025-06-30


