Abstract
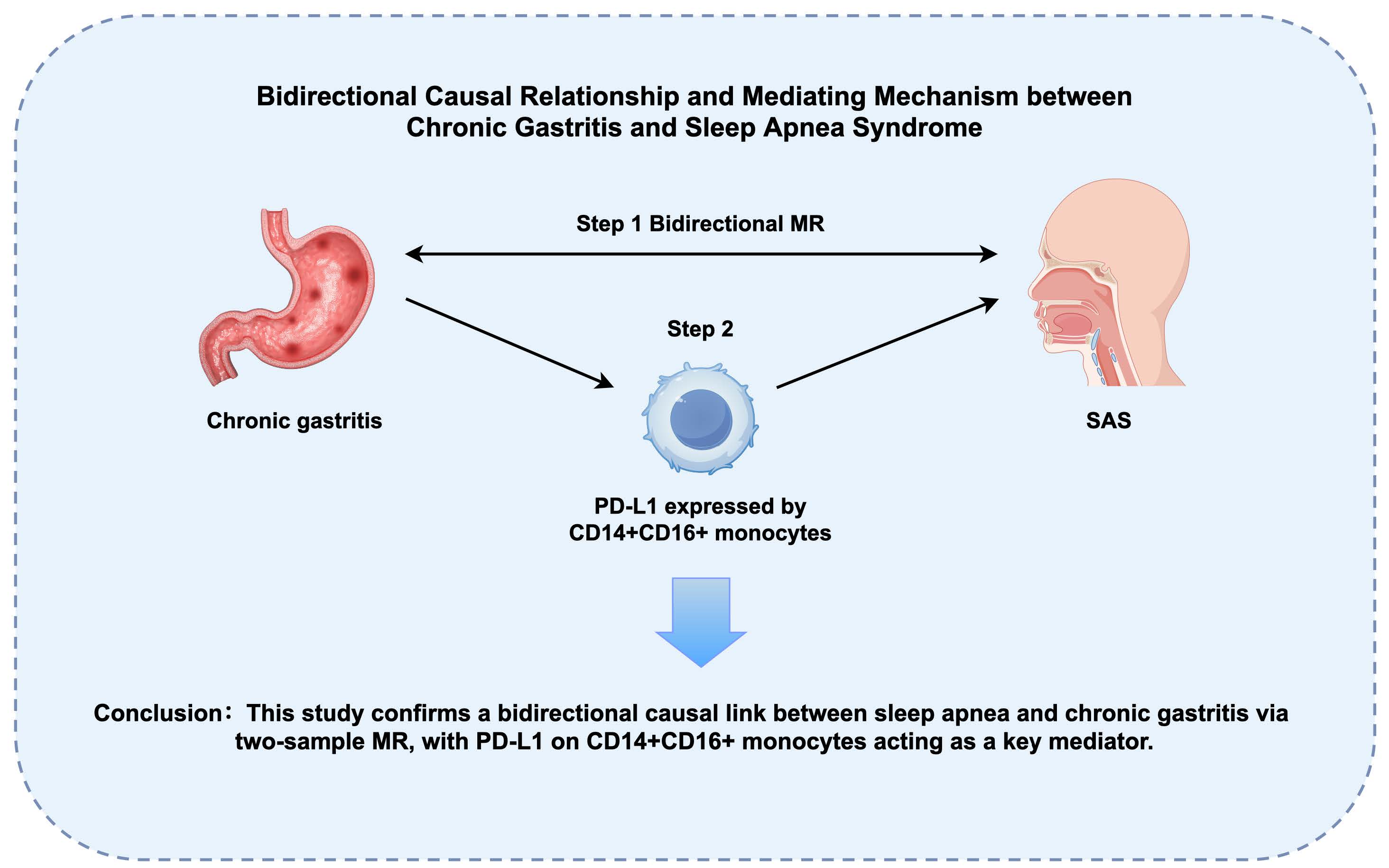
Background: Sleep apnea syndrome (SAS) and chronic gastritis (CG) are prevalent in middle-aged and elderly people. Although clinical observations suggest an association between the two diseases, the causal relationship between them has not been clarified. This study aims to explore the causal relationship between SAS and chronic gastritis and to elucidate the possible mediating mechanisms.
Methods: The causal relationship between SAS and CG was evaluated using five methods (primarily Inverse Variance Weighted (IVW), with others as sensitivity analyses). A two-step Mendelian randomization (MR) study, leveraging GWAS data, was performed to assess the mediating role of signatures in immune cell and inflammatory proteins. Sensitivity analyses were conducted to verify result robustness. The CAUSE framework was applied to address horizontal pleiotropy through comparison of causal and shared genetic models, adjusting for both linkage disequilibrium (LD)-related and independent pleiotropic effects. MR results followed STROBE-MR guidelines.
Results: Through two-sample MR analysis, we identified significant bidirectional causal relationships between sleep apnea syndrome (SAS) and chronic gastritis (forward: OR = 1.131, 95% CI: 1.031-1.240, p = 0.009; reverse: OR = 1.065, 95% CI: 1.007-1.128, p = 0.028). Simulation studies confirmed CAUSE's superior specificity in controlling false positives through its dual-model framework that explicitly accounts for pleiotropic pathways. Two-step MR analysis revealed that the changes in the level of PD-L1 expressed by CD14+CD16+ monocytes played a significant mediating role in the effect of chronic gastritis on SAS.
Conclusion: The bidirectional causal relationship between SAS and chronic gastritis was confirmed through two-sample and two-step MR analyses. Chronic gastritis may increase the risk of SAS through genetic signatures in immune cells, providing new perspectives for future research and aiding in the development of new prevention and treatment strategies.
Keywords:sleep apnea syndrome, chronic gastritis, a two-step Mendelian Randomization Study, a two-sample Mendelian randomization
Introduction
Sleep Apnea Syndrome (SAS) involves repeated episodes of partial or full blockage of the upper airway while sleeping[1,2] , affects millions worldwide and is associated with significant morbidity and mortality[3] . SAS is increasingly prevalent among the middle-aged and elderly, with estimates suggesting a prevalence rate of 20%–40% in people aged[4] . Chronic gastritis is a prevalent, long-term, serious, and subtle illness affecting humans[5] . It is estimated that over half of the global population suffers from this condition to some extent[6] , implying that a significant number of people around the world, possibly in the hundreds of millions, could have chronic gastritis[7] . The intersection of these two conditions presents a fascinating area of study, with potential implications for the diagnosis and management of both disorders.
Despite the increasing prevalence of SAS and the well-documented impact of sleep disorders on gastrointestinal health[8,9] , the specific mechanisms linking SAS to chronic gastritis remain poorly understood. A comprehensive analysis of multiple clinical data and biological mechanisms established a causal link between SAS and chronic gastritis, gastric ulcer, inflammatory bowel disease, and fatty liver[10] , working to elucidate the specific biological pathways involved in the interactions between SAS and gastrointestinal disorders. Another systematic review and Meta-analysis examined the association between SAS and various gastrointestinal disorders (GID), including gastroesophageal reflux disease (GERD) and inflammatory bowel disease (IBD)[11] , which found that GERD is more prevalent in patients with SAS and that these GID disorders may exacerbate the manifestations of SAS. Nevertheless, there is insufficient evidence to support a causal relationship between SAS and chronic gastritis, and its underlying mechanisms remain unclear.
Mendelian randomization (MR) is extensively used in genetic and epidemiological studies, utilizing single nucleotide polymorphisms (SNPs) as natural instrumental variables (IVs) to suggest potential causal links between exposure factors and outcomes[12] . By utilizing identified genetic variations, Mendelian randomization (MR) analysis effectively mitigates the influence of confounding factors and potential reverse causation, thereby providing robust evidence for causal inference. This research employs MR analysis combined with two-step MR to explore the causal relationship between SAS and chronic gastritis, offering new insights into their intricate relationship.
This research aims to investigate the causal relationship between SAS and chronic gastritis. By elucidating the mechanisms connecting SAS and chronic gastritis, it may contribute to understanding the pathogenesis and progression of the disease, as well as identifying potential biomarkers and treatment strategies[13,14] . Further comprehensive research is necessary to understand the exact mechanisms connecting SAS with chronic gastritis.
Methods
Study Design
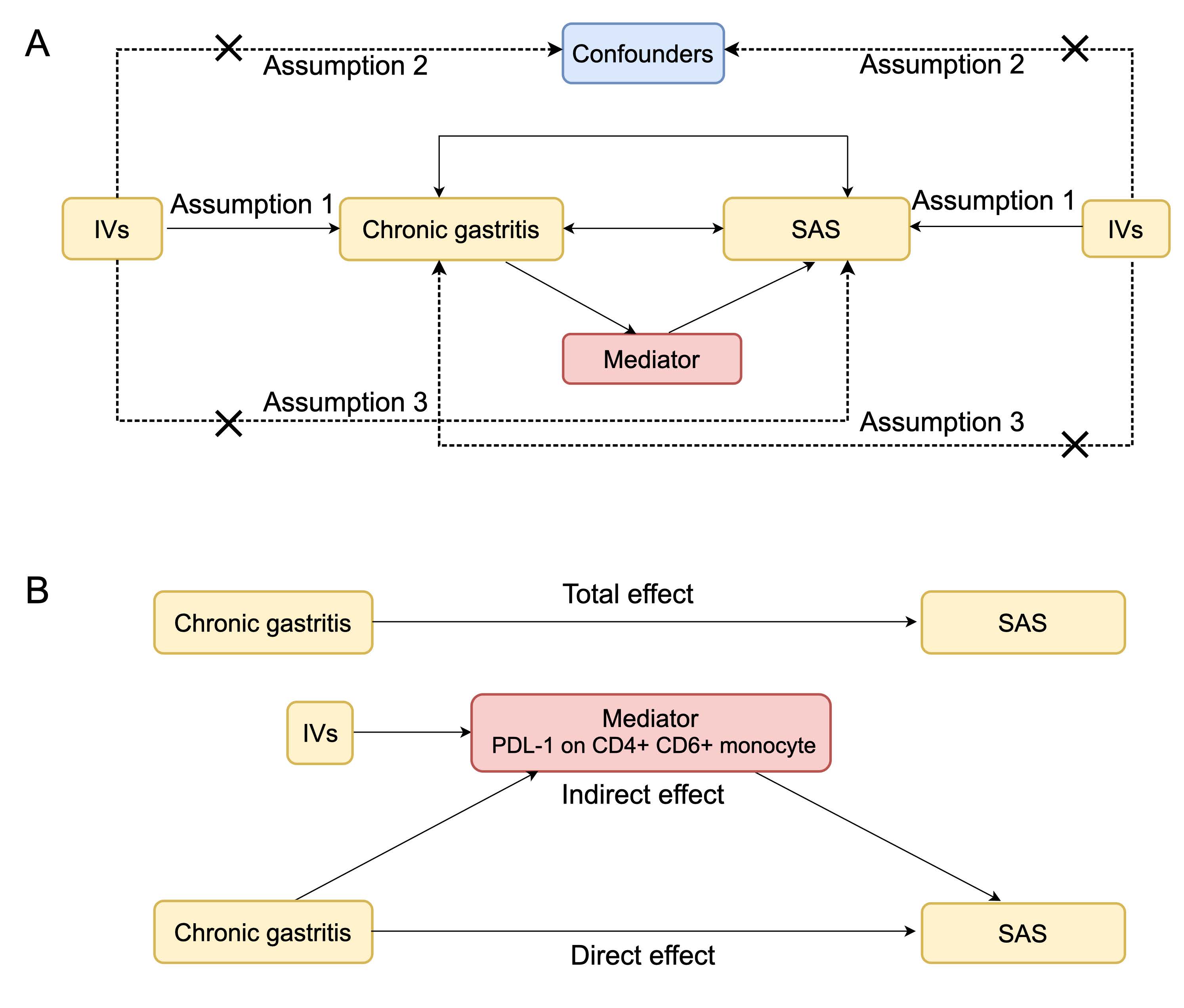
Initially, we conducted a bidirectional two-sample MR analysis to investigate the causal relationships between SAS and the likelihood of developing chronic gastritis. MR-CAUSE was performed to estimate the horizontal pleiotropy in the analysis. Two-step MR analysis was performed to assess the mediating role of signatures in immune cell and inflammatory proteins between the two diseases (Figure 1).
Figure. 1 Flowchart of study design. (A) Three assumptions of MR analysis and (B) the flow of two-step MR analysis.
GWAS data for SAS, chronic gastritis, and signatures in immune cell and inflammatory proteins
The data used in this study were all obtained from publicly available GWAS datasets. The GWAS of the Systemic Autoimmune Syndrome (SAS) included summary statistics (GWAS IDs: finn-b-G6_SLEEEPAPNO, SNPs =16,380,465) from a cohort of 16,761 SAS patients of European descent and 201,194 controls for comparison. The GWAS of chronic gastritis included summary statistics (GWAS IDs: finn-b-K11_CHRONGASTR, SNPs = 16,380,395), with a study sample comprising 5,213 cases and 189,695 controls from Europe desent [15] . The GWAS catalog (GCST90001391 to GCST90002121) provided summary statistics for signatures in immune cell, including data from 3,757 Europeans with 731 signatures in immune cell (SNPs= 22,000,000 approximately)[16] . Information on 91 inflammatory proteins was obtained from GWAS data, which included 14,824 participants of European descent (Zhao et al., 2023)[17] .
Selection of instrument variables
To identify IVs on a global scale, genome-wide significance levels (p < 1e-05) were employed as criteria to screen for IVs that are significantly linked to exposure factors. In all studies, by excluding SNPs within the 10,000 kb and r2 < 0.001 threshold, three high linkage disequilibrium SNPs (rs10938398, rs3996329, rs9937053) were excluded to avoid confounding effects and guarantee the independence of the selected SNPs. These SNPs are strongly associated direct causes of chronic gastritis (visceral fat, obesity BMI, type 2 diabetes, heart disease, neuroticism, depression, caffeine intake, sleep apnea) and have robust clinical evidence necessitating exclusion as confounding factors. Therefore, these confounding factors need to be excluded. Then, the PhenoScanner was used to remove IVs associated with confounders. To avoid weak instrumental bias, the statistical intensity of IV was evaluated using the F-statistic. IVs are considered to have good statistical performance when the F-statistic exceeds 10. The IVs screening procedures and analytical approaches were implemented with reference to methodological frameworks outlined in Burgess et al. [18] and standardized protocols advocated by the International Mendelian Randomization Consortium[19] .
Two-Sample Mendelian Randomization Analysis
All data analysis was performed using the TwoSampleMR software package (version 0.6.8) in R version 4.4.1 (R Foundation for Statistical Computing, Vienna, Austria) for two-sample Mendelian randomization analysis. In this study, the main approach to determine the causal relationship between SAS and chronic gastritis is the inverse variance-weighted method (IVW) using a random-effects model. A range of analysis methods, such as weighted median, MR-Egger, weighted mode, and simple mode methods, were employed to guarantee the reliability of the results and to detect possible horizontal pleiotropies. The IVW method takes into account the uncertainty of the individual SNP effect estimates, giving greater weight to those SNPs with a smaller standard error. MR-Egger explains the potential heterogeneity in IVs and can detect and correct bias to test for horizontal pleiotropy[20,21] . The reliability of causality is enhanced when the results of all methods are roughly the same. This study estimates the causal effect of genetic prediction of SAS on chronic gastritis, represented as odds ratios (OR) and corresponding 95% confidence intervals (CIs). To adjust the p-values, both forward and reverse MR analyses were performed. A p-value of less than 0.05 was considered strong evidence of a causal association. To evaluate the potential sample overlap bias between the GWAS datasets of sleep apnea (SAS) and chronic gastritis, this study employed the Mendelian Randomization with Latent Adjustment for Overlap (MRlap) [22] . This method quantifies the sample overlap ratio through a Bayesian probabilistic model and adjusts the causal effect estimates based on genome-wide genetic correlations. We also use the MR-CAUSE method to avoid false positive results caused by horizontal pleiotropy and strengthen the stable type of positive results[23] . The CAUSE framework was applied to address horizontal pleiotropy through comparison of causal and shared genetic models, adjusting for both LD-related and independent pleiotropic effects. We followed the STROBE-MR (Strengthening the reporting of observational studies in epidemiology using mendelian randomization) guidelines for MR results reporting [24] .
Two-step Mendelian Randomization Analysis
To systematically investigate the bidirectional mechanism of action between chronic gastritis and sleep apnea syndrome (SAS), this study used a two-step MR to characterize their interactions through immune cell features and inflammatory proteins[25] . The first step was to perform MR analysis between chronic gastritis and SAS to obtain the total causal effect. The second step was to evaluate the role of immune cell features or inflammatory proteins in the relationship between chronic gastritis and SAS, respectively. The total effect of chronic gastritis and SAS was decomposed into a direct effect (i.e., the effect of chronic gastritis on SAS without mediation) and an indirect effect (i.e., the effect of chronic gastritis on SAS through mediators). We calculated the effects of chronic gastritis on SAS, with and without the presence of signatures in immune cell and inflammatory proteins, to compare the outcomes and determine the existence of any mediating effects. We conducted inverse variance-weighted (IVW) analysis to evaluate the causal effects before and after the removal of intermediaries. Additionally, the weighted median method and MR-Egger were performed as complementary methods, and MR-Egger was used to identify and adjust for bias to assess potential horizontal pleiotropy, thereby reducing bias caused by horizontal pleiotropy [20] . We also performed a reverse pathway analysis, in which SAS was used as an exposure to identify immune cell signatures and inflammatory protein markers of its effects, and thus assessed the direct and indirect effects of SAS on chronic gastritis.
Sensitivity analysis
After the preliminary screening, we evaluated the heterogeneity of the Cochran’s Q test of IVW and MR-Egger methods for the significant results, and when p < 0.05, significant heterogeneity was considered to be present [26] . Horizontal pleiotropy was evaluated using the MR-Egger regression intercept and MR-PRESSO. A p-value greater than 0.05 for the MR-Egger regression intercept suggests that horizontal pleiotropy can be disregarded. For MR-PRESSO, p > 0.05 after overall inspection shows that horizontal pleiotropy is negligible [27] . Repeat the analysis after eliminating the peripheral SNP, and compare the differences before and after deleting the outlier. To assess the stability of the results, a leave-one-out analysis was conducted by excluding each SNP individually, and the results were considered stable if the exclusion of any single SNP did not lead to significant changes [28] . Funnel plots were used to illustrate the distinct relationships between SNPs and were employed to evaluate bias in the findings. Reverse MR can assess if the genetic variation serving as an exposure trait also has a causal relationship with the outcome, affecting it via the exposure trait.
To validate the robustness of the main Mendelian Randomization (MR) analysis results, this study utilized three external datasets UK Biobank verification: ukb-b-6716 (n=342,000), ukb-b-12191 (n=408,000), and ukb-d-K11_CHRONGASTR (n=28,500). All validation cohorts had no overlap with the main samples, and the phenotype definitions were complementary to reduce bias from a single data source. We retained the instrumental selected in the main (p < 1e-05, F-statistic > 10) and removed SNPs in the validation cohorts that exhibited high linkage disequilibrium (LD) with the main analysis SNPs (10,000 kb, r2 < 0.001), while also excluding SNPs with inconsistent exposure association directions. Statistical methods employed the inverse-variance weighted (IVW) method as the primary estimator, supplemented by the weighted median method and MR-Egger regression (testing for horizontal pleiotropy), with outlier SNPs removed using MR-PRESSO. Consistency was crossvalidated through IVW, MR-Egger, weighted median, and pleiotropy tests (e.g., MR-PRESSO), and heterogeneity was assessed using Cochran’s Q (p < 0.05).
Through cross-validation using inverse variance weighted (IVW), MR-Egger regression, weighted median, and MR-PRESSO methods, we systematically assess the robustness of results. The statistical assumptions of different methods are mutually independent (e.g., IVW assumes no pleiotropy of instrumental variables, while MR-Egger allows for directional pleiotropy), and the consistency in effect direction and significance can effectively replace traditional multiple corrections [29] . The International Mendelian Randomization Study Guidelines indicate that when the main analysis (IVW) is consistent with sensitivity methods (such as weighted median, MR-Egger), additional corrections may be overly conservative and obscure true associations [30] . Several MR studies [30,31] in similar designs did not use BH correction, instead relying on inter-method consistency as a robustness standard [31,32] .
MR-PRESSO was performed in R (version 4.4.1) using the MR-PRESSO software package (version 1.0), while the "forestplot" software package was used for graphical representation.
Results
Causal Relationship between Chronic Gastritis and Sleep Apnea Syndrome
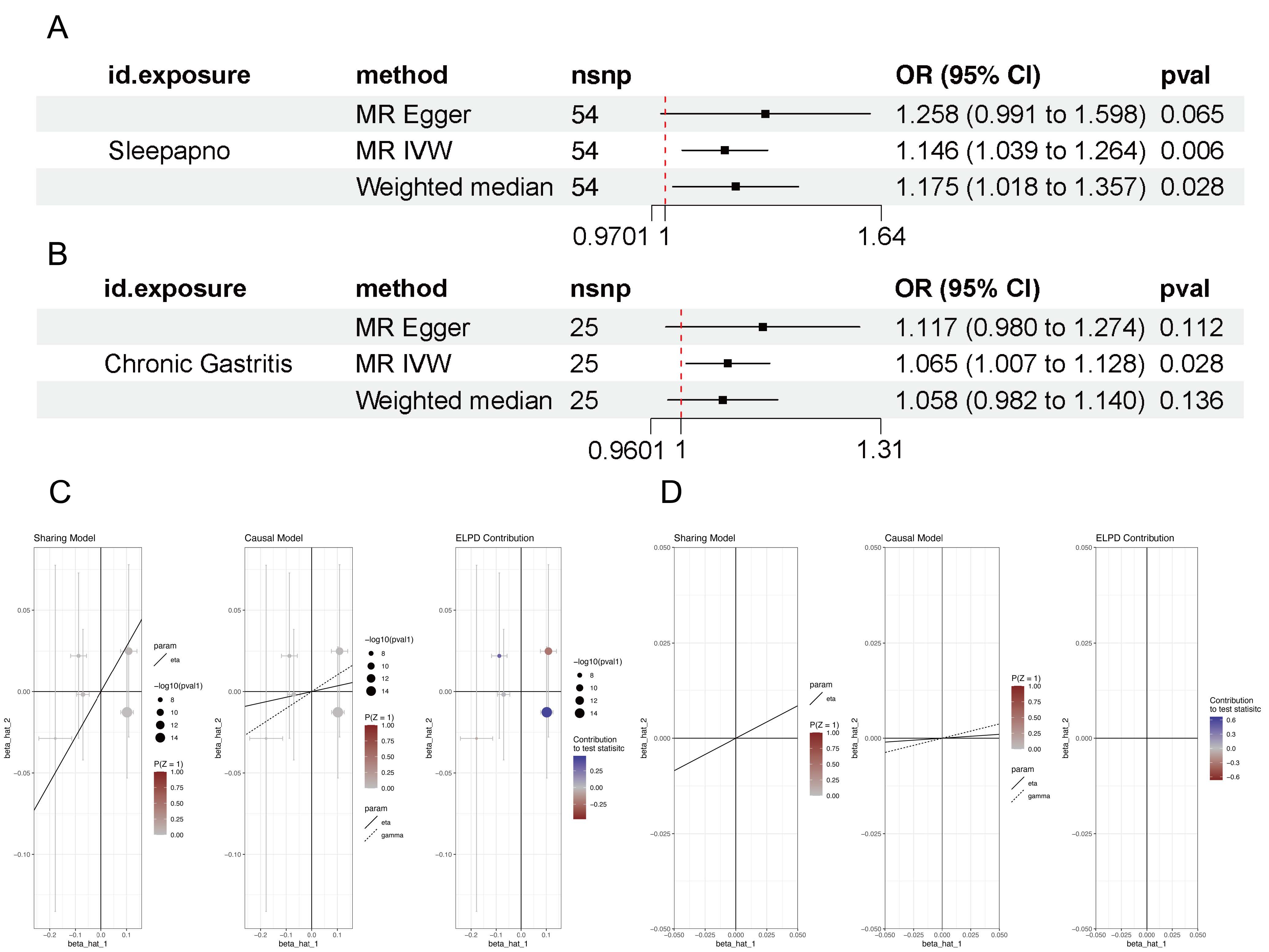
In a two-sample MR analysis, the IVW method analysis identified a notable association between SAS and an higher risk of chronic gastritis (OR = 1.131, 95% CI: 1.031-1.240, p = 0.009)(Figure. S1, Table S1). We systematically screened confounding factors associated with chronic gastritis and SAS, and the PhenoScanner database was used to eliminate confounder-related SNPs. SNPs (rs10938398, rs3996329, rs9937053) significantly related to confounding factors of chronic gastritis (diabetes, excessive coffee drinking, heavy alcohol consumption, long-term heavy smoking, non-steroidal anti-inflammatory drugs) were removed [33] , but no SNPs significantly related to confounding factors were found in reverse MR. To test the robustness of the results, we also used 4 tests, including MR-Egger regression, weighted median method, simple mode method, and weighted mode method, and the positive causal relationship between SAS and chronic gastritis remained significant (Figure 2a). Then, we conducted a comprehensive and sufficient sensitivity test. In the heterogeneity test analysis, the P value of the Cochran’s Q test was greater than 0.05, indicating the absence of significant heterogeneity (Table S2); the leave-one-out analysis further confirmed the stability of the primary outcome, indicating that no individual SNP overinfluenced the effect estimate (Figure. S2); and the funnel plot indicated that the analysis was not significantly biased (Figure. S3). To exclude the effect of horizontal pleiotropy, we performed an MR-Egger regression cut-off distance term test and an MR-PRESSO test (Table S3). The results showed that there was no significant horizontal pleiotropy (p > 0.05). The results of MR-Cause analysis showed that the causal model was superior to the shared model (delta_elpd < 0), and this method showed negative results under the causal model (p > 0.05) (Figure 2c), and the results were robust. In order to comprehensively assess the relationship between SAS and chronic gastritis, we used two-way MR to assess the inverse effect of chronic gastritis on SAS levels to explore whether chronic gastritis may also be a contributing factor of SAS. IVW results showed that chronic gastritis was associated with an increase in SAS levels (OR = 1.065, 95% CI: 1.007-1.128, p = 0.028) (Figure 2b) (Table S4). The sensitive test of reverse Mendelian randomization and the MR-CAUSE test have the same steps as before, and the results are stable without heterogeneity, horizontal pleiotropy, or bias. The above results indicate that there is a bidirectional causal relationship between SAS and chronic gastritis. The leave-one-out method and funnel plots both suggested that the data were reliable (Supplementary file). MRlap analysis revealed partial sample overlap between the SAS and chronic gastritisAS datasets (both derived from the FinnGen biobank), but its impact on causal effect estimates was statistically insignificant (forward: p_different = 0.688, reverse: p_different = 0.576), indicating that the research conclusions are not affected by overlap. Detailed analysis results are provided in the supplementary materials. This study validates the robustness of the main analysis results based on three independent subsets from the UK Biobank (ukb-b-6716, ukb-b-12191, ukb-d-K11_CHRONGASTR), all of which have no overlap with the main analysis sample and have complementary phenotype definitions. However, none of the three external datasets achieved statistical significance, yet we still believe that the main analysis results are scientifically reasonable: There are currently no other available datasets for sleep apnea syndrome (SAS), and the dataset for gastritis is relatively small with insufficient SNP numbers, but our original results have been validated through MRlap and various sensitivity tests, making them reliable.
Figure. 2 (a) The forest plot of the potential positive causal relationship between SAS and chronic gastritis. (b) The forest plot of the potential reverse causal relationship between SAS and chronic gastritis. MR: mendelian randomization. Results of MR-Cause Analysis. (c) MR-Cause test of chronic gastritis caused by SAS. (d) MR-Cause test of SAS caused by chronic gastritis. The p-values from the sensitivity analysis are uncorrected for validation, and their consistency supports the robustness of the results.
Mediating signatures in immune cell and inflammatory proteins in chronic gastritis triggering SAS
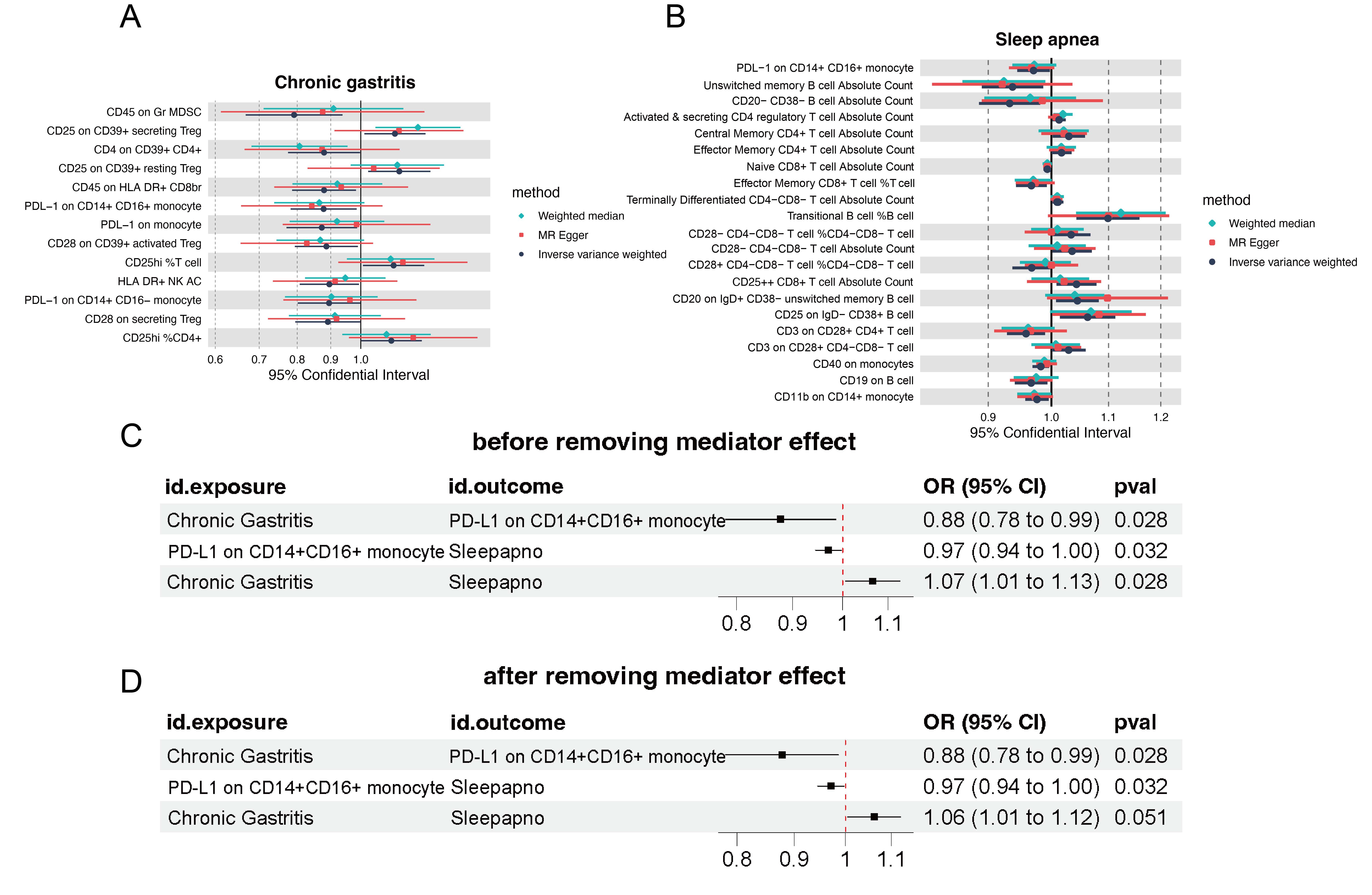
Chronic gastritis increases the risk of SAS mediated by PDL-1 on CD14+CD16+ monocytes
Next, we explored the causal relationship between chronic gastritis and genetic markers of signatures in immune cell (Figure 3a) and between genetic markers of signatures in immune cell and SAS (Figure 3b) through three methods: MR Egger, IVW, and weighted median. The results were significant, indicating that chronic gastritis can increase the risk of SAS by changing genetic signatures in immune cell, especially PD-L1 expression on CD14+CD16+ monocytes. Finally, we estimated the direct effect of chronic gastritis on SAS after accounting for mediation via signatures in immune cell and inflammatory proteins (before removing: OR = 1.07, 95% CI: 1.01-1.13, p = 0.028; after removing: OR = 1.06, 95% CI: 1.01-1.12, p = 0.051) (Figure 3c) and found that the significance after removal of the immunomediator was significantly attenuated, revealing that chronic gastritis may increase the risk of SAS by upregulating PD-L1 levels on CD14+CD16+ monocytes. To exclude horizontal pleiotropy, we performed the MR-Egger regression intercept term test and the MR-PRESSO test, which showed that there was no significant horizontal pleiotropy (p > 0.05). In the heterogeneity test, the P-value of Cochran’s Q was greater than 0.05, indicating the absence of significant heterogeneity. Leave-one analysis showed that no single SNP overinfluenced the effect estimate, further confirming the stability of the results. Through the above methods, it is shown that the changes of genetic markers of signatures in immune cell constitute a significant intermediary pathway between chronic gastritis and SAS, and chronic gastritis can increase the risk of SAS by activating immunity.
Figure. 3 Mendelian Randomization Study for mediation analysis of genetic signatures in immune cell. (a) Causal relationship between chronic gastritis and genetic signatures in immune cell. (b) Causal relationship between genetic signatures in immune cell and SAS. (c) and (d) Mediation Mendelian randomization of PDL-1 on CD14+CD16+ monocytes. Two forest plots reveal the causal relationship between chronic gastritis and SNS before and after removing PDL-1 on CD14+CD16+ monocytes.
No inflammatory protein were found to mediate chronic gastritis and SAS
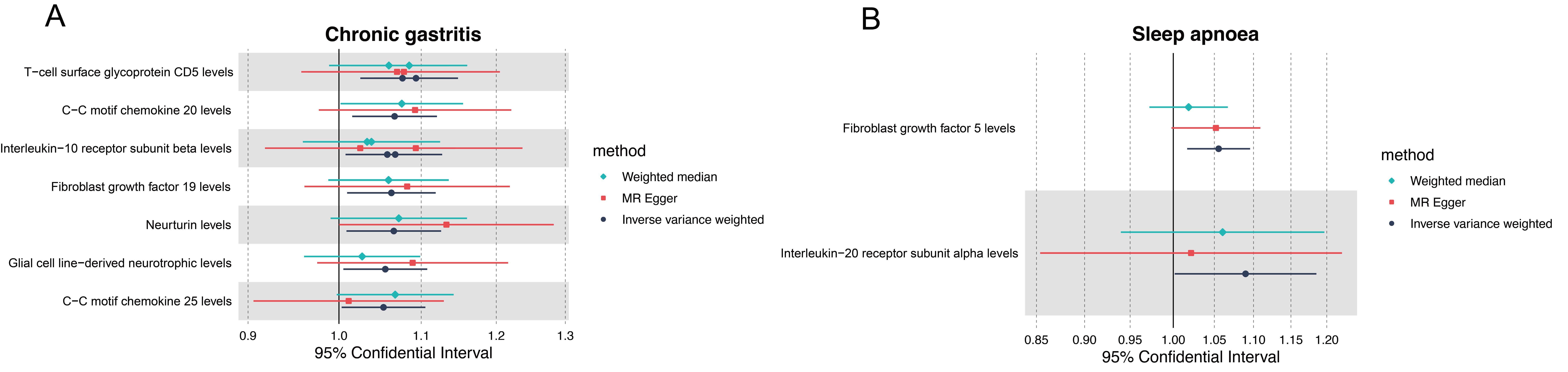
In further MR for mediation analysis, we also explored the causal relationship between the level of inflammatory proteins caused by chronic gastritis (Figure 4a) and the causal relationship between the level of inflammatory proteins leading to SAS (Figure 4b). The results showed that chronic gastritis caused T-cell surface glycoprotein CD5 levels, T-cell surface glycoprotein CD6 isoform levels, C-C motif chemokine 20 levels, Interleukin-10 receptor subunit alpha levels, Fibroblast growth factor 19 levels, Neurturin levels, Interleukin-10 receptor subunit beta levels, Glial cell line-derived neurotrophic factor levels, and C-C motif chemokine 25 levels. These nine inflammatory proteins increased. Fibroblast growth factor 5 levels and Interleukin-20 receptor subunit alpha levels are positively correlated with the incidence of SAS, but in our study sample, no specific inflammatory proteins was found to significantly mediate the relationship between chronic gastritis and SAS. This finding suggests that future research may pay more attention to exploring the non-inflammatory link between chronic gastritis and SAS and how to improve the comorbidity of the two diseases through comprehensive interventions.
Figure. 4 Mendelian randomization study for mediation analysis of inflammatory proteins. (a) Causal relationship between chronic gastritis and inflammatory proteins. (b) Causal relationship between inflammatory proteins and SAS
Mediating signatures in immune cell and inflammatory proteins in SAS triggering chronic gastritis
In exploring the mediating pathways of SAS on chronic gastritis (CG), candidate inflammatory proteins (C-C motif chemokine 19 levels, Natural killer cell receptor 2B4 levels) and immune cell markers (CD11c+ monocyte %monocyte, etc.) did not reach statistical significance. Sensitivity analyses still indicate this null association. This suggests that there are no apparent immune inflammatory mediators mediating the effect of SAS on chronic gastritis, and other unmeasured mechanisms may exist.
Discussion
Sleep Apnea Syndrome (SAS) is a widespread disorder characterized by disruptions in breathing during sleep, manifested through recurrent episodes of complete (apnea) or partial (hypopnea) cessation of airflow, resulting in intermittent oxygen deprivation[33-35] . This condition is associated with symptoms such as intermittent hypoxemia, sleep fragmentation, systemic inflammatory responses, oxidative stress, and disruptions in physiological homeostasis[33] . Moreover, the inadequate oxygen supply can lead to dysbiosis of the intestinal microbiota, alterations in intestinal metabolites[36] , and damage to the intestinal barrier due to insufficient mucosal oxygenation[37] . These symptoms may contribute to an increased incidence of chronic gastritis. Conversely, nocturnal awakenings, sleep deprivation, and sleep fragmentation induced by chronic gastritis may exacerbate the occurrence of SAS[38,39] and impair the recruitment of upper airway dilators, potentially leading to further obstructive respiratory events[40] . Considering the limitations associated with time ranking in existing observational studies, this research elucidated the potential causal relationship between SAS and chronic gastritis as risk factors, utilizing a two-way, two-sample MR approach. Additionally, the study investigated the mechanistic pathways mediating the interaction between these conditions through two-step MR analysis.
Notably, our findings revealed, for the first time, that chronic gastritis mediates an increased risk of SAS via the expression of PD-L1 by CD14+CD16+ monocytes. CD14+CD16+ monocytes contribute to SAS-associated oxidative stress and induce low-grade systemic inflammation by infiltrating the endothelium and overexpressing PD-L1[41-43] . Furthermore, they inhibit the activation, proliferation, and cytotoxic activity of CD8+ T cells through the hypoxia-dependent PD-L1/PD-1 pathway[41,44] . The primary rationale for the efficacy of low hypoglossal nerve stimulation therapy in treating SAS lies in its ability to markedly decrease the expression of PD-L1 and the proportion of CD16+ monocytes, which have notable pro-inflammatory effects. Additionally, it reduces the expression levels of TNF-α and IL-1β[45] . In the context of chronic gastritis, gastric dendritic cells located in the submucosa and intrinsic mucosa frequently express substantial amounts of PD-L1, thereby creating a cellular barrier that prevents T cells from infiltration and erosion of the gastric mucosa[46] . Similarly, monocytes may be extensively recruited and express PD-L1 in response to inflammatory stimuli, potentially exacerbating the severity of SAS. Nonetheless, there is currently no research indicating that gastritis influences SAS through PD-L1 expression by CD14+CD16+ monocytes, and the specific mechanisms involved remain to be elucidated.
This study presents several advantages, notably the utilization of association analysis based on clinical cross-sectional studies, which further elucidates the bidirectional causal relationship between SAS and chronic gastritis through MR analysis. This approach effectively circumvents the ethical concerns associated with observational analyses, as well as the confounding effects and biases introduced by reverse causality. Furthermore, two-step MR was employed to investigate the potential mediation effect between SAS and chronic gastritis. This analysis confirmed the involvement of 91 inflammatory proteins and 731 signatures in immune cell, thereby establishing a foundation for future research endeavors.
Nevertheless, this study possesses certain limitations that warrant discussion. Firstly, although our investigation did not identify any guided pleiotropy, the possibility of potential pleiotropy remains, which is a common challenge in MR research and can introduce bias. Secondly, the MR study was performed on individuals of European descent, while the cross-sectional study involved a multi-ethnic Chinese cohort. Additionally, the GWAS sample size for chronic gastritis was relatively small. Future research focusing on a homogeneous ethnic group is necessary to mitigate potential confounding effects arising from population heterogeneity. Lastly, our study is essentially a statistical analysis and lacks experimental validation using blood samples from chronic gastritis.
Conclusion
This study proved that there is a significant bidirectional causal relationship between SAS and chronic gastritis by two-sample MR. MR study for mediation analysis found that the level of PD-L1 expressed by CD14+CD16+ monocytes plays a key mediating role between chronic gastritis and SAS. These findings provide potential targets for the development of novel therapeutic strategies for chronic gastritis and SAS, and in particular, interventions targeting PD-L1 may help reduce the risk of both diseases.[1,2]
Abbreviations
95% CIs: 95% confidence intervals; CAUSE: Causal Analysis Using Summary Effect Estimates; CG: Chronic gastritis; GERD: Gastroesophageal reflux disease; GID: Gastrointestinal disorders; GWAS: Genome-wide association study; IBD: Inflammatory bowel disease; IVs: Instrumental variables; IVW: the Inverse Variance Weighted; LD: Linkage disequilibrium; MR:Mendelian Randomization Study; MRlap: the Mendelian Randomization with Latent Adjustment for Overlap; OR: Odds ratios; SAS: Sleep apnea syndrome; SNPs: Single nucleotide polymorphisms.
Supplementary Material
Supplementary methods, results, spectra, figures.
Declarations
Acknowledgements
Not applicable.
Author contributions
Ziyan Zhao: Methodology. Data curation, Writing - original draft, Writing - review & editing. Yanyu Zhu: Writing - review & editing. Chang You: Visualization. Yan Wang: Writing - review & editing. Tianchi Zhuang: Writing - review & editing: Conceptualization. Yingqi Yang: Conceptualization, Funding acquisition, Writing - review & editing. All authors read and approved the final manuscript
Ethics approval and consent to participate
Not applicable.
Funding information
Not applicable.
Competing Interests
The authors declare that they have no existing or potential commercial or financial relationships that could create a conflict of interest at the time of conducting this study.
Data Availability
This study analyzed datasets that are publicly accessible. These datasets can be found at the following URLs: FinnGen (https://storage.googleapis.com/finngen-public-data-r9/ summary_stats/finngen_R9_M13_OSTEONECROSIS.gz) and GWAS Catalog (https://www.ebi.ac.uk/gwas/downloads/ summary-statistics).
References
[1] Wanyan P, et al. Obstructive sleep apnea hypopnea syndrome: Protocol for the development of a core outcome set. Medicine (Baltimore). 2020;99(34):e21591. DOI: 10.1097/md.0000000000021591.
[2] Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. The Report of an American Academy of Sleep Medicine Task Force. Sleep. 1999;22(5):667-89.
[3] Piccirillo F, et al. A State-of-the-Art Review on Sleep Apnea Syndrome and Heart Failure. Am J Cardiol. 2023;195:57-69. DOI: 10.1016/j.amjcard.2023.02.020.
[4] Peppard PE, et al. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol. 2013;177(9):1006-14. DOI: 10.1093/aje/kws342.
[5] Guidelines for diagnosis and treatment of chronic gastritis in China (2022, Shanghai). J Dig Dis. 2023;24(3):150-80. DOI: 10.1111/1751-2980.13193.
[6] Du Y, et al. Chronic gastritis in China: a national multi-center survey. BMC Gastroenterol. 2014;14:21. DOI: 10.1186/1471-230x-14-21.
[7] Sipponen P, Maaroos HI. Chronic gastritis. Scand J Gastroenterol. 2015;50(6):657-67. DOI: 10.3109/00365521.2015.1019918.
[8] Orr WC, et al. The effect of sleep on gastrointestinal functioning in common digestive diseases. Lancet Gastroenterol Hepatol. 2020;5(6):616-24. DOI: 10.1016/s2468-1253(19)30412-1.
[9] Vege SS, et al. Functional gastrointestinal disorders among people with sleep disturbances: a population-based study. Mayo Clin Proc. 2004;79(12):1501-6. DOI: 10.4065/79.12.1501.
[10] Zhang Z, et al. Investigating the causal links between obstructive sleep apnea and gastrointestinal diseases mediated by metabolic syndrome through mendelian randomization. Sci Rep. 2024;14(1):26247. DOI: 10.1038/s41598-024-77471-x.
[11] Xia W, et al. Relationship between obstructive sleep apnoea syndrome and essential hypertension: a dose-response meta-analysis. Sleep Med. 2018;47:11-18. DOI: 10.1016/j.sleep.2018.03.016.
[12] Lawlor DA, et al. Mendelian randomization: using genes as instruments for making causal inferences in epidemiology. Stat Med. 2008;27(8):1133-63. DOI: 10.1002/sim.3034.
[13] Mashaqi S, Gozal D. Obstructive Sleep Apnea and Systemic Hypertension: Gut Dysbiosis as the Mediator? J Clin Sleep Med. 2019;15(10):1517-27. DOI: 10.5664/jcsm.7990.
[14] Lu D, et al. Profiling of lung microbiota in the patients with obstructive sleep apnea. Medicine (Baltimore). 2018;97(26):e11175. DOI: 10.1097/md.0000000000011175.
[15] Sun M, et al. Causal relationships of Helicobacter pylori and related gastrointestinal diseases on Type 2 diabetes: Univariable and Multivariable Mendelian randomization. PLoS One. 2024;19(4):e0300835. DOI: 10.1371/journal.pone.0300835.
[16] Li W, et al. Complex causal association between genetically predicted 731 immunocyte phenotype and osteonecrosis: a bidirectional two-sample Mendelian randomization analysis. Int J Surg. 2024;110(6):3285-93. DOI: 10.1097/js9.0000000000001327.
[17] Xiao C, et al. Two-sample Mendelian randomization analysis of 91 circulating inflammatory protein levels and amyotrophic lateral sclerosis. Front Aging Neurosci. 2024;16:1367106. DOI: 10.3389/fnagi.2024.1367106.
[18] Burgess S, et al. Guidelines for performing Mendelian randomization investigations: update for summer 2023. Wellcome Open Res. 2019;4:186. DOI: 10.12688/wellcomeopenres.15555.3.
[19] Davey Smith G, Hemani G. Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Hum Mol Genet. 2014;23(R1):R89-98. DOI: 10.1093/hmg/ddu328.
[20] Verbanck M, et al. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet. 2018;50(5):693-8. DOI: 10.1038/s41588-018-0099-7.
[21] Bowden J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol. 2015;44(2):512-25. DOI: 10.1093/ije/dyv080.
[22] Mounier N, Kutalik Z. Bias correction for inverse variance weighting Mendelian randomization. Genet Epidemiol. 2023;47(4):314-31. DOI: 10.1002/gepi.22522.
[23] Morrison J, et al. Mendelian randomization accounting for correlated and uncorrelated pleiotropic effects using genome-wide summary statistics. Nat Genet. 2020;52(7):740-7. DOI: 10.1038/s41588-020-0631-4.
[24] Skrivankova VW, et al. Strengthening the reporting of observational studies in epidemiology using mendelian randomisation (STROBE-MR): explanation and elaboration. Bmj. 2021;375:n2233. DOI: 10.1136/bmj.n2233.
[25] Carter AR, et al. Mendelian randomisation for mediation analysis: current methods and challenges for implementation. Eur J Epidemiol. 2021;36(5):465-78. DOI: 10.1007/s10654-021-00757-1.
[26] Hemani G, Bowden J, Davey Smith G. Evaluating the potential role of pleiotropy in Mendelian randomization studies. Hum Mol Genet. 2018;27(R2):R195-208. DOI: 10.1093/hmg/ddy163.
[27] Bowden J, et al. Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet Epidemiol. 2016;40(4):304-14. DOI: 10.1002/gepi.21965.
[28] Hu J, et al. Reverse causal relationship between periodontitis and shortened telomere length: Bidirectional two-sample Mendelian random analysis. Front Immunol. 2022;13:1057602. DOI: 10.3389/fimmu.2022.1057602.
[29] Burgess S, Thompson SG. Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol. 2017;32(5):377-89. DOI: 10.1007/s10654-017-0255-x.
[30] Hemani G, et al. The MR-Base platform supports systematic causal inference across the human phenome. Elife. 2018;7. DOI: 10.7554/eLife.34408.
[31] Larsson SC, et al. Genetic predisposition to smoking in relation to 14 cardiovascular diseases. Eur Heart J. 2020;41(35):3304-10. DOI: 10.1093/eurheartj/ehaa193.
[32] Kurki MI, et al. FinnGen provides genetic insights from a well-phenotyped isolated population. Nature. 2023;613(7944):508-18. DOI: 10.1038/s41586-022-05473-8.
[33] Yan W, et al. Obstructive sleep apnea and 19 gastrointestinal diseases: a Mendelian randomization study. Front Psychiatry. 2024;15:1256116. DOI: 10.3389/fpsyt.2024.1256116.
[34] Ma B, et al. Association Between Abdominal Adipose Tissue Distribution and Obstructive Sleep Apnea in Chinese Obese Patients. Front Endocrinol (Lausanne). 2022;13:847324. DOI: 10.3389/fendo.2022.847324.
[35] Antonaglia C, et al. Low arousal threshold: a common pathophysiological trait in patients with obstructive sleep apnea syndrome and asthma. Sleep Breath. 2023;27(3):933-41. DOI: 10.1007/s11325-022-02665-4.
[36] Hatamnejad MR, et al. Selective serotonin reuptake inhibitors and inflammatory bowel disease; Beneficial or malpractice. Front Immunol. 2022;13:980189. DOI: 10.3389/fimmu.2022.980189.
[37] Wu J, et al. Disrupted intestinal structure in a rat model of intermittent hypoxia. Mol Med Rep. 2016;13(5):4407-13. DOI: 10.3892/mmr.2016.5068.
[38] Gold AR, Gold MS. Con: Sleep fragmentation causes hypersomnolence in OSA. Sleep Med Rev. 2021;55:101399. DOI: 10.1016/j.smrv.2020.101399.
[39] Liu H, et al. Effect of Helicobacter pylori-Associated Chronic Gastritis on Autonomous Activity and Sleep Quality in Mice. Front Pharmacol. 2022;13:785105. DOI: 10.3389/fphar.2022.785105.
[40] Chiba S. 0700 Prediction Of Surgical Outcome Using Respiratory Pattern Clafification. Sleep. 2020;43(Supplement_1):A267-A267. DOI: 10.1093/sleep/zsaa056.696 %J Sleep.
[41] Mya HT, et al. PD-1 and PD-L1 Are Overexpressed in the "Intermediate CD14+CD16+" and "Non Classical CD14lowCD16+" but Not in the "Classical CD14+CD16-" Monocytes in the Peripheral Blood of Chronic Myelomonocytic Leukemia. Blood. 2015;126(23):1694-1694. DOI: 10.1182/blood.V126.23.1694.1694 %J Blood.
[42] Bolasco P, et al. Could there be Haemodynamic Stress Effects on Pro-Inflammatory CD14+CD16+ Monocytes during Convective-Diffusive Treatments? A Prospective Randomized Controlled Trial. Blood Purif. 2019;47(4):385-94. DOI: 10.1159/000494711.
[43] Ziegler-Heitbrock L. The CD14+ CD16+ blood monocytes: their role in infection and inflammation. J Leukoc Biol. 2007;81(3):584-92. DOI: 10.1189/jlb.0806510.
[44] Costa F, et al. PD-L1/PD-1 Pattern of Expression Within the Bone Marrow Immune Microenvironment in Smoldering Myeloma and Active Multiple Myeloma Patients. Front Immunol. 2020;11:613007. DOI: 10.3389/fimmu.2020.613007.
[45] Johnson MD, et al. Model-based analysis of implanted hypoglossal nerve stimulation for the treatment of obstructive sleep apnea. Sleep. 2021;44(44 Suppl 1):S11-s19. DOI: 10.1093/sleep/zsaa269.
[46] Go DM, et al. Programmed Death Ligand 1-Expressing Classical Dendritic Cells MitigateHelicobacter-Induced Gastritis. Cell Mol Gastroenterol Hepatol. 2021;12(2):715-39. DOI: 10.1016/j.jcmgh.2021.04.007.
 Figures
Figures References
References Peer
Peer Information
InformationFigure. 1 Flowchart of study design. (A) Three assumptions of MR analysis and (B) the flow of two-step MR analysis.

Figure. 2 (a) The forest plot of the potential positive causal relationship between SAS and chronic gastritis. (b) The forest plot of the potential reverse causal relationship between SAS and chronic gastritis. MR: mendelian randomization. Results of MR-Cause Analysis. (c) MR-Cause test of chronic gastritis caused by SAS. (d) MR-Cause test of SAS caused by chronic gastritis. The p-values from the sensitivity analysis are uncorrected for validation, and their consistency supports the robustness of the results.

Figure. 3 Mendelian Randomization Study for mediation analysis of genetic signatures in immune cell. (a) Causal relationship between chronic gastritis and genetic signatures in immune cell. (b) Causal relationship between genetic signatures in immune cell and SAS. (c) and (d) Mediation Mendelian randomization of PDL-1 on CD14+CD16+ monocytes. Two forest plots reveal the causal relationship between chronic gastritis and SNS before and after removing PDL-1 on CD14+CD16+ monocytes.

Figure. 4 Mendelian randomization study for mediation analysis of inflammatory proteins. (a) Causal relationship between chronic gastritis and inflammatory proteins. (b) Causal relationship between inflammatory proteins and SAS

[1] Wanyan P, et al. Obstructive sleep apnea hypopnea syndrome: Protocol for the development of a core outcome set. Medicine (Baltimore). 2020;99(34):e21591. DOI: 10.1097/md.0000000000021591.
[2] Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. The Report of an American Academy of Sleep Medicine Task Force. Sleep. 1999;22(5):667-89.
[3] Piccirillo F, et al. A State-of-the-Art Review on Sleep Apnea Syndrome and Heart Failure. Am J Cardiol. 2023;195:57-69. DOI: 10.1016/j.amjcard.2023.02.020.
[4] Peppard PE, et al. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol. 2013;177(9):1006-14. DOI: 10.1093/aje/kws342.
[5] Guidelines for diagnosis and treatment of chronic gastritis in China (2022, Shanghai). J Dig Dis. 2023;24(3):150-80. DOI: 10.1111/1751-2980.13193.
[6] Du Y, et al. Chronic gastritis in China: a national multi-center survey. BMC Gastroenterol. 2014;14:21. DOI: 10.1186/1471-230x-14-21.
[7] Sipponen P, Maaroos HI. Chronic gastritis. Scand J Gastroenterol. 2015;50(6):657-67. DOI: 10.3109/00365521.2015.1019918.
[8] Orr WC, et al. The effect of sleep on gastrointestinal functioning in common digestive diseases. Lancet Gastroenterol Hepatol. 2020;5(6):616-24. DOI: 10.1016/s2468-1253(19)30412-1.
[9] Vege SS, et al. Functional gastrointestinal disorders among people with sleep disturbances: a population-based study. Mayo Clin Proc. 2004;79(12):1501-6. DOI: 10.4065/79.12.1501.
[10] Zhang Z, et al. Investigating the causal links between obstructive sleep apnea and gastrointestinal diseases mediated by metabolic syndrome through mendelian randomization. Sci Rep. 2024;14(1):26247. DOI: 10.1038/s41598-024-77471-x.
[11] Xia W, et al. Relationship between obstructive sleep apnoea syndrome and essential hypertension: a dose-response meta-analysis. Sleep Med. 2018;47:11-18. DOI: 10.1016/j.sleep.2018.03.016.
[12] Lawlor DA, et al. Mendelian randomization: using genes as instruments for making causal inferences in epidemiology. Stat Med. 2008;27(8):1133-63. DOI: 10.1002/sim.3034.
[13] Mashaqi S, Gozal D. Obstructive Sleep Apnea and Systemic Hypertension: Gut Dysbiosis as the Mediator? J Clin Sleep Med. 2019;15(10):1517-27. DOI: 10.5664/jcsm.7990.
[14] Lu D, et al. Profiling of lung microbiota in the patients with obstructive sleep apnea. Medicine (Baltimore). 2018;97(26):e11175. DOI: 10.1097/md.0000000000011175.
[15] Sun M, et al. Causal relationships of Helicobacter pylori and related gastrointestinal diseases on Type 2 diabetes: Univariable and Multivariable Mendelian randomization. PLoS One. 2024;19(4):e0300835. DOI: 10.1371/journal.pone.0300835.
[16] Li W, et al. Complex causal association between genetically predicted 731 immunocyte phenotype and osteonecrosis: a bidirectional two-sample Mendelian randomization analysis. Int J Surg. 2024;110(6):3285-93. DOI: 10.1097/js9.0000000000001327.
[17] Xiao C, et al. Two-sample Mendelian randomization analysis of 91 circulating inflammatory protein levels and amyotrophic lateral sclerosis. Front Aging Neurosci. 2024;16:1367106. DOI: 10.3389/fnagi.2024.1367106.
[18] Burgess S, et al. Guidelines for performing Mendelian randomization investigations: update for summer 2023. Wellcome Open Res. 2019;4:186. DOI: 10.12688/wellcomeopenres.15555.3.
[19] Davey Smith G, Hemani G. Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Hum Mol Genet. 2014;23(R1):R89-98. DOI: 10.1093/hmg/ddu328.
[20] Verbanck M, et al. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet. 2018;50(5):693-8. DOI: 10.1038/s41588-018-0099-7.
[21] Bowden J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol. 2015;44(2):512-25. DOI: 10.1093/ije/dyv080.
[22] Mounier N, Kutalik Z. Bias correction for inverse variance weighting Mendelian randomization. Genet Epidemiol. 2023;47(4):314-31. DOI: 10.1002/gepi.22522.
[23] Morrison J, et al. Mendelian randomization accounting for correlated and uncorrelated pleiotropic effects using genome-wide summary statistics. Nat Genet. 2020;52(7):740-7. DOI: 10.1038/s41588-020-0631-4.
[24] Skrivankova VW, et al. Strengthening the reporting of observational studies in epidemiology using mendelian randomisation (STROBE-MR): explanation and elaboration. Bmj. 2021;375:n2233. DOI: 10.1136/bmj.n2233.
[25] Carter AR, et al. Mendelian randomisation for mediation analysis: current methods and challenges for implementation. Eur J Epidemiol. 2021;36(5):465-78. DOI: 10.1007/s10654-021-00757-1.
[26] Hemani G, Bowden J, Davey Smith G. Evaluating the potential role of pleiotropy in Mendelian randomization studies. Hum Mol Genet. 2018;27(R2):R195-208. DOI: 10.1093/hmg/ddy163.
[27] Bowden J, et al. Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet Epidemiol. 2016;40(4):304-14. DOI: 10.1002/gepi.21965.
[28] Hu J, et al. Reverse causal relationship between periodontitis and shortened telomere length: Bidirectional two-sample Mendelian random analysis. Front Immunol. 2022;13:1057602. DOI: 10.3389/fimmu.2022.1057602.
[29] Burgess S, Thompson SG. Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol. 2017;32(5):377-89. DOI: 10.1007/s10654-017-0255-x.
[30] Hemani G, et al. The MR-Base platform supports systematic causal inference across the human phenome. Elife. 2018;7. DOI: 10.7554/eLife.34408.
[31] Larsson SC, et al. Genetic predisposition to smoking in relation to 14 cardiovascular diseases. Eur Heart J. 2020;41(35):3304-10. DOI: 10.1093/eurheartj/ehaa193.
[32] Kurki MI, et al. FinnGen provides genetic insights from a well-phenotyped isolated population. Nature. 2023;613(7944):508-18. DOI: 10.1038/s41586-022-05473-8.
[33] Yan W, et al. Obstructive sleep apnea and 19 gastrointestinal diseases: a Mendelian randomization study. Front Psychiatry. 2024;15:1256116. DOI: 10.3389/fpsyt.2024.1256116.
[34] Ma B, et al. Association Between Abdominal Adipose Tissue Distribution and Obstructive Sleep Apnea in Chinese Obese Patients. Front Endocrinol (Lausanne). 2022;13:847324. DOI: 10.3389/fendo.2022.847324.
[35] Antonaglia C, et al. Low arousal threshold: a common pathophysiological trait in patients with obstructive sleep apnea syndrome and asthma. Sleep Breath. 2023;27(3):933-41. DOI: 10.1007/s11325-022-02665-4.
[36] Hatamnejad MR, et al. Selective serotonin reuptake inhibitors and inflammatory bowel disease; Beneficial or malpractice. Front Immunol. 2022;13:980189. DOI: 10.3389/fimmu.2022.980189.
[37] Wu J, et al. Disrupted intestinal structure in a rat model of intermittent hypoxia. Mol Med Rep. 2016;13(5):4407-13. DOI: 10.3892/mmr.2016.5068.
[38] Gold AR, Gold MS. Con: Sleep fragmentation causes hypersomnolence in OSA. Sleep Med Rev. 2021;55:101399. DOI: 10.1016/j.smrv.2020.101399.
[39] Liu H, et al. Effect of Helicobacter pylori-Associated Chronic Gastritis on Autonomous Activity and Sleep Quality in Mice. Front Pharmacol. 2022;13:785105. DOI: 10.3389/fphar.2022.785105.
[40] Chiba S. 0700 Prediction Of Surgical Outcome Using Respiratory Pattern Clafification. Sleep. 2020;43(Supplement_1):A267-A267. DOI: 10.1093/sleep/zsaa056.696 %J Sleep.
[41] Mya HT, et al. PD-1 and PD-L1 Are Overexpressed in the "Intermediate CD14+CD16+" and "Non Classical CD14lowCD16+" but Not in the "Classical CD14+CD16-" Monocytes in the Peripheral Blood of Chronic Myelomonocytic Leukemia. Blood. 2015;126(23):1694-1694. DOI: 10.1182/blood.V126.23.1694.1694 %J Blood.
[42] Bolasco P, et al. Could there be Haemodynamic Stress Effects on Pro-Inflammatory CD14+CD16+ Monocytes during Convective-Diffusive Treatments? A Prospective Randomized Controlled Trial. Blood Purif. 2019;47(4):385-94. DOI: 10.1159/000494711.
[43] Ziegler-Heitbrock L. The CD14+ CD16+ blood monocytes: their role in infection and inflammation. J Leukoc Biol. 2007;81(3):584-92. DOI: 10.1189/jlb.0806510.
[44] Costa F, et al. PD-L1/PD-1 Pattern of Expression Within the Bone Marrow Immune Microenvironment in Smoldering Myeloma and Active Multiple Myeloma Patients. Front Immunol. 2020;11:613007. DOI: 10.3389/fimmu.2020.613007.
[45] Johnson MD, et al. Model-based analysis of implanted hypoglossal nerve stimulation for the treatment of obstructive sleep apnea. Sleep. 2021;44(44 Suppl 1):S11-s19. DOI: 10.1093/sleep/zsaa269.
[46] Go DM, et al. Programmed Death Ligand 1-Expressing Classical Dendritic Cells MitigateHelicobacter-Induced Gastritis. Cell Mol Gastroenterol Hepatol. 2021;12(2):715-39. DOI: 10.1016/j.jcmgh.2021.04.007.
Peer-review Terminology
Identity transparency: Single anonymized
Reviewer interacts with: Editor
Details
This is an open access article under the terms of the Creative Commons Attribution License(http://creativecommons.org/licenses/by/4.0/), which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Publication History
Received 2025-01-25
Accepted 2025-05-18
Published 2025-06-30


